Summary: Researchers at Cornell University have discovered a new pathway that involves immune cells which may contribute to the development of Alzheimer’s disease.
Source: Cornell University
By studying the effects of immune cells that surround blood vessels in the brain, Weill Cornell Medicine researchers have discovered a new pathway involving these cells that may contribute to the cause of Alzheimer’s disease.
One of the hallmarks of Alzheimer’s disease is the accumulation of plaque deposits, or abnormal protein fragments, from a peptide called amyloid-beta. Amyloid-beta destroys neurons and damages brain blood vessels with the help of highly reactive molecules, called free radicals, which are derived from oxygen. For their study, published May 17 in Circulation Research, the researchers sought to determine which cells in the brain were responsible for producing the free radicals, also known as reactive oxygen species (ROS).
“These free radicals are nasty molecules that paralyze the vessels so that they can’t function normally. As a result, brain cells are deprived of the blood supply of oxygen and glucose they need to function properly,” said senior study author Dr. Costantino Iadecola, director of the Feil Family Brain and Mind Research Institute and the Anne Parrish Titzell Professor of Neurology at Weill Cornell Medicine.
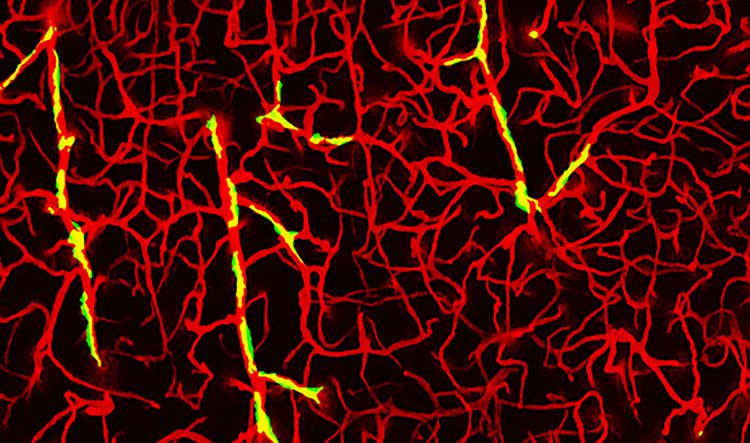
In their study, the investigators examined cells in the brain called perivascular macrophages (PVM), which surround blood vessels and pick up and remove damaging metabolic byproducts floating around the brain. The team focused on these cells because amyloid-beta tends to accumulate in the vascular space where PVM are located.
“We saw that when cells are exposed to amyloid beta, these PVM start producing free radicals like crazy,” Iadecola said.
To determine whether the PVM were involved in the ROS production, the researchers removed PVM by injecting into the brains of mice spherical lipid droplets called liposomes that contained the drug clodronate.
“The liposomes acted as a kind of Trojan horse,” said Laibaik Park, assistant professor of research in neuroscience in the Feil Family Brain and Mind Research Institute, and first author of the study. “The PVM digest the lipids and sets the drug loose into the cell, forcing them to die.”
Park and Iadecola administered the liposomes in mice with Alzheimer’s disease; ROS paralyzed the blood vessels in the rodents. Remarkably, the team found that the blood vessels in these mice appeared to work normally once rid of PVM, despite the presence of amyloid-beta. “This showed that without the PVM, amyloid-beta was unable to exert its damaging effects on brain blood vessels,” Park said.
The researchers then sought to learn what caused the PVM to produce ROS. Previous studies showed that macrophages have innate immune receptors on their surface called CD36 that help them bind and interact with other immune cells and proteins. The next step was to determine what happens when CD36 was not present, Iadecola said.
To remove the CD36 receptors from PVM, the researchers first used radiation to destroy the PVM. Then, old PVM were replaced through a bone marrow transplant with new PVM that did not have the receptor.
“When we did that, it was as good as getting rid of the whole macrophage, proving that the presence of CD36 on PVM was necessary for the ROS production and vascular damage to occur,” Iadecola said.
The researchers ultimately discovered that when amyloid beta binds to the CD36 receptor of PVM, a signaling pathway is triggered, which causes an enzyme called NADPH oxidase to produce free radicals.
“It was previously thought that amyloid-beta was going to work its way from the brain into the wall of blood vessels, reaching endothelial cells, which would be the source of the radicals,” Iadecola said. “But in reality, amyloid-beta globs onto the macrophages to activate them. This makes perfect sense because endothelial cells are inside the vessels away from brain amyloid-beta, while PVM are outside the vessel continuously bathed by amyloid-beta coming from the brain.”
Genetic studies have long suggested that the brain’s innate immune cells, like microglia and macrophages, contribute to Alzheimer’s disease, but how these cells damaged the brain was not known. “Our study identifies a novel way in which immune cells could contribute to Alzheimer’s disease, and provide a new therapeutic approach to suppress their damaging effects,” Iadecola said.
Researchers will now seek to develop methods that block CD36 or NADPH oxidase in the cells, which may reduce the amount of amyloid-beta in the brain and potentially slow the progression of Alzheimer’s disease. “Delaying the process may give you more years with a healthy mind,” Iadecola said.
He also added that the maintenance of blood vessel health is crucial for a wide variety of brain diseases. “Our research shows that even in a disease that doesn’t start with a blood vessel, by making the vessels work better, there may be a beneficial effect,” Iadecola said. “Beside Alzheimer’s disease, our study provides us with a new way to look at vascular risk factors that cause stroke and dementia, like hypertension and diabetes, because in these conditions these PVM cells were not previously considered as major players.”
Source: Molly Schulson – Cornell University
Image Source: NeuroscienceNews.com image is credited to the researchers.
Original Reserch: Abstract for “Brain Perivascular Macrophages Initiate the Neurovascular Dysfunction of Alzheimer Aβ Peptides” by Laibaik Park, Ken Uekawa, Lidia Garcia-Bonilla, Kenzo Koizumi, Michelle Murphy, Rose Pitstick, Linda H Younkin, Steven G Younkin, Ping Zhou, Geroge A Carlson, Josef Anrather, and Costantino Iadecola in Circulation Research. Published online May 17 doi:10.1161/CIRCRESAHA.117.311054
[cbtabs][cbtab title=”MLA”]Cornell University “Purpose in Life by Day Linked to Better Sleep at Night.” NeuroscienceNews. NeuroscienceNews, 12 July 2017.
<https://neurosciencenews.com/alzheimers-immune-cells-7069/>.[/cbtab][cbtab title=”APA”]Cornell University (2017, July 12). Purpose in Life by Day Linked to Better Sleep at Night. NeuroscienceNew. Retrieved July 12, 2017 from https://neurosciencenews.com/alzheimers-immune-cells-7069/[/cbtab][cbtab title=”Chicago”]Cornell University “Purpose in Life by Day Linked to Better Sleep at Night.” https://neurosciencenews.com/alzheimers-immune-cells-7069/ (accessed July 12, 2017).[/cbtab][/cbtabs]
Abstract
Brain Perivascular Macrophages Initiate the Neurovascular Dysfunction of Alzheimer Aβ Peptides
Rationale: Increasing evidence indicates that alterations of the cerebral microcirculation may play a role in Alzheimer’s disease (AD), the leading cause of late-life dementia. The amyloid-β peptide (Aβ), a key pathogenic factor in AD, induces profound alterations in neurovascular regulation through the innate immunity receptor CD36, which, in turn, activates a Nox2-containing NADPH oxidase leading to cerebrovascular oxidative stress. Brain perivascular macrophages (PVM) located in the perivascular space, a major site of brain Aβ collection and clearance, are juxtaposed to the wall of intracerebral resistance vessels and are a powerful source of reactive oxygen species (ROS).
Objective: We tested the hypothesis that PVM are the main source of ROS responsible for the cerebrovascular actions of Aβ, and that CD36 and Nox2 in PVM are the molecular substrates of the effect.
Methods and Results: Selective depletion of PVM using intracerebroventricular injection of clodronate abrogates the ROS production and cerebrovascular dysfunction induced by Aβ applied directly to the cerebral cortex, administered intravascularly or overproduced in the brain of transgenic mice expressing mutated forms of the amyloid precursor protein (Tg2576 mice). In addition, using bone marrow chimeras we demonstrate that PVM are the cells expressing CD36 and Nox2 responsible for the dysfunction. Thus, deletion of CD36 or Nox2 from PVM abrogates the deleterious vascular effects of Aβ, whereas wild-type PVM reconstitute the vascular dysfunction in CD36-null mice.
Conclusions: The data identify PVM as a previously unrecognized effector of the damaging neurovascular actions of Aβ and unveil a new mechanism by which brain-resident innate immune cells and their receptors may contribute to the pathobiology of AD.
“Brain Perivascular Macrophages Initiate the Neurovascular Dysfunction of Alzheimer Aβ Peptides” by Laibaik Park, Ken Uekawa, Lidia Garcia-Bonilla, Kenzo Koizumi, Michelle Murphy, Rose Pitstick, Linda H Younkin, Steven G Younkin, Ping Zhou, Geroge A Carlson, Josef Anrather, and Costantino Iadecola in Circulation Research. Published online May 17 doi:10.1161/CIRCRESAHA.117.311054






