Summary: Researchers have discovered that two genetic variants, APOE4 and TREM2 R47H, together cause a harmful inflammatory response in brain immune cells, particularly in females. This response leads to more severe brain damage, including tau protein clumps, which are linked to Alzheimer’s disease progression.
The study showed that female mice with these variants experienced increased inflammation in brain immune cells, known as microglia, which become dysfunctional and activate the cGAS-STING pathway. These findings suggest that targeting immune pathways in a sex-specific manner may be a promising strategy for Alzheimer’s treatment and prevention.
Key Facts:
- APOE4 and TREM2 variants together cause harmful brain inflammation in females.
- Microglia dysfunction in females with these variants accelerates Alzheimer’s damage.
- Targeting immune pathways may offer new, sex-specific Alzheimer’s treatments.
Source: Weill Cornell University
Weill Cornell Medicine investigators have found that two genetic variants that confer high risk of Alzheimer’s disease (AD) together trigger a harmful inflammatory response in the brain’s immune cells, particularly in females, in a preclinical model.
The findings, published Sept. 30 in Neuron, emphasize the importance of considering sex differences in Alzheimer’s research – a step that could ultimately lead to more precise and effective treatments.
AD affects millions of people worldwide, with women disproportionately impacted – nearly twice as many females develop the disease compared with males. To advance therapeutic approaches, investigators are trying to determine the basis for these differences in vulnerability.
“Previous studies have shown that a gene variant called APOE4 increases AD risk more in women compared with men. The current study zeroed in on cellular activities that go awry when APOE4 and a variant of the TREM2 gene, which also raises AD risk, are present together in females. Because the proteins encoded by these genes have a variety of functions in cells, it has been unclear how the particular variants contribute to a vulnerability to the disease.
“Although these are two of the strongest risk factors for AD, little is known about how they enhance disease risk and they have not been often studied together,” said senior author Dr. Li Gan, director of the Helen and Robert Appel Alzheimer’s Disease Research Institute and the Burton P. and Judith B. Resnick Distinguished Professor in Neurodegenerative Diseases in the Feil Family Brain and Mind Research Institute at Weill Cornell Medicine.
“Our goal was to combine these risk factors to highlight what pathways are altered when the risk of disease is strongest.”
Dr. Gan and her team, including lead author Dr. Gillian Carling, a graduate student in the Weill Cornell Medicine Graduate School of Medical Sciences at the time of the study, established mouse models for AD carrying human versions of APOE4 and TREM2 R47H, a rare variant that increases AD risk 2-4.5-fold.
The mice also carried a mutation that leads to the development of clumps of tau protein—abundant in AD brains and closely associated with cognitive decline in patients. The team examined the mice at 9-10 months of age, roughly equivalent to middle age in humans, to assess how these genetic variants impacted brain health.
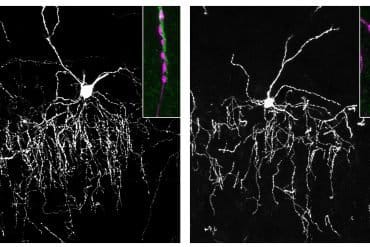
They found that female, but not male, mice with both APOE4 and TREM2 R47H exhibited significant damage to the brain region that plays an important role in thinking and memory. This damage included more severe tau protein clumps compared with those mice without these gene combinations.
The researchers attributed the damage in these female brains to the brain’s immune cells, called microglia. Normally, microglia protect the brain, but in this case, they became “senescent”, referring to aged cells that lost their ability to function properly.
Instead of cleaning up damaged cells and protein clumps, these aged microglia linger around and release inflammatory chemicals through a pathway known as cGAS-STING. Notably, the study found that these harmful effects were more pronounced in female mice, a finding that aligns with reports that APOE4 poses a stronger risk for women than men.
“Our research showed that when the two Alzheimer’s risk factors are combined in females with tau aggregates, the cGAS-STING pathway becomes highly activated,” Dr. Carling said. Suppressing this deleterious pathway lowered harmful inflammatory factors and rescued the aging phenotype in microglia.
The study underscores the need to consider sex differences in Alzheimer’s research and treatment, the researchers say, as the disease may progress differently in men and women and therefore may require tailored approaches, commented Dr. Gan.
By identifying the role of immune pathways like cGAS-STING in the progression of Alzheimer’s, particularly in individuals with high-risk genetic variants, the researchers hope to open the door to new strategies for treatment—and potentially prevention.
Funding: This work was supported by the National Institute on Aging, part of the National Institutes of Health, through grant numbers R01AG076448, R01AG072758, R01AG054214, R01AG074541, R01AG064239, K99AG078493, and F31AG079560.
About this genetics, neurology, and inflammation research news
Author: Krystle Lopez
Source: Weill Cornell University
Contact: Krystle Lopez – Weill Cornell University
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Alzheimer’s disease-linked risk alleles elevate microglial cGAS-associated senescence and neurodegeneration in a tauopathy model” by Li Gan et al. Neuron
Abstract
Alzheimer’s disease-linked risk alleles elevate microglial cGAS-associated senescence and neurodegeneration in a tauopathy model
The strongest risk factors for late-onset sporadic Alzheimer’s disease (AD) include the ε4 allele of apolipoprotein E (APOE), the R47H variant of triggering receptor expressed on myeloid cells 2 (TREM2), and female sex.
Here, we combine APOE4 and TREM2R47H (R47H) in female P301S tauopathy mice to identify the pathways activated when AD risk is the strongest, thereby highlighting detrimental disease mechanisms.
We find that R47H induces neurodegeneration in 9- to 10-month-old female APOE4 tauopathy mice.
The combination of APOE4 and R47H (APOE4-R47H) worsened hyperphosphorylated tau pathology in the frontal cortex and amplified tauopathy-induced microglial cyclic guanosine monophosphate (GMP)-AMP synthase (cGAS)-stimulator of interferon genes (STING) signaling and downstream interferon response.
APOE4-R47H microglia displayed cGAS- and BAX-dependent upregulation of senescence, showing association between neurotoxic signatures and implicating mitochondrial permeabilization in pathogenesis.
By uncovering pathways enhanced by the strongest AD risk factors, our study points to cGAS-STING signaling and associated microglial senescence as potential drivers of AD risk.