Summary: A pairing of presynaptic and postsynaptic activity in a population of hippocampal neurons can reduce the required number of synaptic plasticity evoking events. When spikes occur within ten milliseconds of each other, synapses strengthen for up to four hours.
Source: National University of Singapore, Yong Loo Lin School of Medicine
Remember hearing people being called slow learners by teachers and parents? That oft-used description of someone who takes a wee bit longer to process information now has a scientific basis for its existence.
Scientists from the NUS Yong Loo Lin School of Medicine have found that the rapidity with which a person is able to grasp, process, understand, store and use information comes down to the speed and timing with which the neurons in the brain fire off. The closer the gap between the firing of one neuron and the next, the greater the speed with which the information is received, stored and acted upon.
In other words, when it comes to quick thinking, timing makes all the difference.
In making this discovery, Dr. Sajikumar Sreedharan and undergraduate student Karen Pang, Ph.D. student Mahima Sharma, post-doctoral fellow Dr. Krishna Kumar — all from the Department of Physiology — and collaborator Dr. Thomas Behnisch from China, provided fundamental information about the precise timing that can critically affect the formation of memory processes. The study was published in the prestigious international journal Proceedings of the National Academy of Sciences of the United States of America (PNAS) on 01 March 2019.
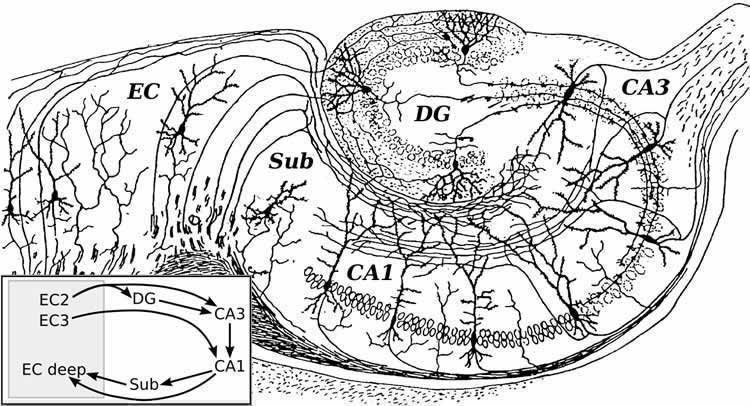
The capacity to adapt and learn with experience is one of the most intriguing features of the human brain. This fascinating organ is composed of billions of neurons, which are in turn connected to many other cells. The physical connections between neurons, called synapses, are where neurons communicate with each other. Synapses are remarkably plastic — these connections can strengthen or weaken dynamically in response to incoming information. Such changes in the efficacy of the synapses underlie learning and the formation of memory in the brain.
The NUS team found that neurons in the hippocampus, a brain region critical for the formation of memory, use a surprisingly wide variety of learning mechanisms. One such form of learning, termed “spike-timing-dependent plasticity (STDP),” depends on the timing of each pair of electrical spikes (electrical activity used to transmit information within neurons) in the pre-synaptic neuron and the post-synaptic neuron. An electrical spike in the presynaptic neuron stimulates the neuron to release neurotransmitters, which travel across the synapse to activate the postsynaptic neuron, where the information is converted back into an electrical spike. When the pre- and postsynaptic neurons are active at the same time (less than 30 milliseconds apart), the connections between them are strengthened. However, when the presynaptic neuron fires earlier by 30 milliseconds or more, or when the postsynaptic neuron fires earlier by more than 10 milliseconds, the connections are strengthened to a lesser degree.
In addition, the researchers demonstrated that when the pre- and postsynaptic spikes occur at the same time, the increase in synaptic strength persists for several hours, and the synapse can even strengthen weak information so that it gets stored. The effect was specific, working only to strengthen this synapse, not to enhance changes in other synapses. This study reveals how important the split-second timing in neuronal activity is in shaping information processing in the brain.
The researchers could detect the longer-term effects of the inter-spike timing because they studied the synaptic changes for a longer duration (4 hours) than the durations employed in previous studies, which were typically less than 1.5 hours.
The STDP model has been proposed to explain spatial and temporal sequence learning. Moreover, the STDP model comes in handy in various situations where object identification and decision-making have to be done quickly, e.g. projectile avoidance or friend-foe identification. For instance, when a ball is flying towards us, we have to identify the object and its trajectory of travel within a very small window of time, in order to swiftly take action to avoid being hit. Similarly, when we come across another person, we have to quickly decide whether they are a friend or a foe. And this recognition requires the coordinated action of various areas of the brain within an activity timing window explained by STDP.
“Unfortunately, the ability for the brain to change in response to such precise timing of information flow may be lacking in brains affected by Alzheimer’s Disease as the hippocampus is particularly damaged in this common cause of dementia. This study may provide the foundation for understanding how such timing differences alter brain function and also how these changes could be reversed or mitigated. This might enable clinicians to help patients who suffer from memory loss,” said Dr. Christopher Chen, Senior Consultant Neurologist, National University Hospital and Director of the Memory Aging and Cognition Centre, National University Health System.
A comprehensive understanding of the factors that shape neural connections is critical for our understanding of information processing in the brain. It also helps us to understand how memories are formed. Furthermore, a firm grasp of these neural computational rules can help guide the building of artificial intelligence technology, e.g. deep neural networks, which are inspired by the brain’s learning mechanisms.
“In the case of Autism Spectrum Disorder, some of the neural systems are more active than the others. This could be the reason why some autistic people are good at certain tasks like arts or mathematics but have difficulty socializing. Using artificial intelligence, it might be possible to identify the neural networks that are more or less active and it might be possible to normalize their functioning using STDP rules,” said Dr Sajikumar.
Based on this improved understanding of how normal brains compute information and learn, researchers can identify mechanisms for further study that may be involved in conditions like schizophrenia, depression, sleep loss, stroke, chronic pain, learning disability, and Alzheimer’s disease.
Source:
National University of Singapore, Yong Loo Lin School of Medicine
Media Contacts:
Gerry Everding – National University of Singapore, Yong Loo Lin School of Medicine
Image Source:
The image is in the public domain.
Original Research: Closed access
Karen Ka Lam Pang, Mahima Sharma, Kumar Krishna-K., Thomas Behnisch, Sreedharan Sajikumar. “Long-term population spike-timing-dependent plasticity promotes synaptic tagging but not cross-tagging in rat hippocampal area CA1.” Proceedings of the National Academy of Sciences, 2019; 116 (12): 5737 doi:10.1073/pnas.1817643116
Abstract
Long-term population spike-timing-dependent plasticity promotes synaptic tagging but not cross-tagging in rat hippocampal area CA1
In spike-timing-dependent plasticity (STDP), the direction and degree of synaptic modification are determined by the coherence of pre- and postsynaptic activities within a neuron. However, in the adult rat hippocampus, it remains unclear whether STDP-like mechanisms in a neuronal population induce synaptic potentiation of a long duration. Thus, we asked whether the magnitude and maintenance of synaptic plasticity in a population of CA1 neurons differ as a function of the temporal order and interval between pre- and postsynaptic activities. Modulation of the relative timing of Schaffer collateral fibers (presynaptic component) and CA1 axons (postsynaptic component) stimulations resulted in an asymmetric population STDP (pSTDP). The resulting potentiation in response to 20 pairings at 1 Hz was largest in magnitude and most persistent (4 h) when presynaptic activity coincided with or preceded postsynaptic activity. Interestingly, when postsynaptic activation preceded presynaptic stimulation by 20 ms, an immediate increase in field excitatory postsynaptic potentials was observed, but it eventually transformed into a synaptic depression. Furthermore, pSTDP engaged in selective forms of late-associative activity: It facilitated the maintenance of tetanization-induced early long-term potentiation (LTP) in neighboring synapses but not early long-term depression, reflecting possible mechanistic differences with classical tetanization-induced LTP. The data demonstrate that a pairing of pre- and postsynaptic activities in a neuronal population can greatly reduce the required number of synaptic plasticity-evoking events and induce a potentiation of a degree and duration similar to that with repeated tetanization. Thus, pSTDP determines synaptic efficacy in the hippocampal CA3–CA1 circuit and could bias the CA1 neuronal population toward potentiation in future events.