A team of Bio-X scientists applied microscopy know-how to a long-standing theory in neuroscience: if brain connections called synapses store memories, those synapses should last as long as the memories themselves. It turns out they do, as Mark Schnitzer was able to show.
Our memories are as fleeting as the brain structures that store them, or so the theory goes. When the connections – called synapses – between neurons break, the memories they hold are thought to evaporate along with them.
The idea seemed good, but has been hard to test. Now a Stanford team has taken on the challenge, studying a brain region called the hippocampus, which stores “episodic” memories. These are the memories of events or conversations that might be forgotten over time if the memories aren’t used. The challenge to studying synapses in this region is that the hippocampus is so deep and the connections so densely packed that no microscope could easily monitor the synapses’ longevity.
Now Mark Schnitzer, an associate professor of biology and of applied physics, has leveraged microscopy tools developed in his lab and for the first time was able to monitor the connections, called synapses, between hippocampal neurons and confirm what neuroscientists thought might be happening. In the mice he and his team studied, the connections between neurons lasted about 30 days, roughly the duration over which episodic memories are believed to stay in the mouse hippocampus. The work was published on June 22 in Nature.
“Just because the community has had a longstanding idea, that doesn’t make it right,” Schnitzer said. Now that the idea has been validated, he said, his technique could open up new areas of memory research: “It opens the door to the next set of studies, such as memory storage in stress or disease models.”
Mobile memories
When mice experience a new episode or learn a new task that requires spatial navigation, the memory is stored for about a month in a structure at the center of the brain called the hippocampus (it is stored slightly longer in people). If mice have hippocampus-disrupting surgery within a month of forming a memory – a memory of meeting a new cage-mate or navigating a maze – that memory is lost.
If the disruption occurs after more than a month, then the mouse still retains the memory of a new friend or location of food. That’s because the memory had been relocated to a different region of the brain, the neocortex, and is no longer susceptible to disruption in the hippocampus.
“The thought is that memories are gradually moved around the brain,” said Schnitzer, who is also a member of Stanford Bio-X and the Stanford Neurosciences Institute.
“The neocortex is a long-term repository, whereas considerable evidence indicates that memories stay in the mouse hippocampus only about a month.”
In the past, scientists at Cold Spring Harbor Laboratory in New York and elsewhere had monitored connections between neurons in the neocortex, nearer the brain’s surface and therefore visible with little disruption to the brain. They watched not the connections themselves, but the bulbous projections called spines that form connections at their tips. Watching the spines come and go serves as a proxy for knowing when excitatory connections between neurons are created and broken.
Those scientists found that about half of the spines in the neocortex were permanent and the rest turned over approximately every five to 15 days.
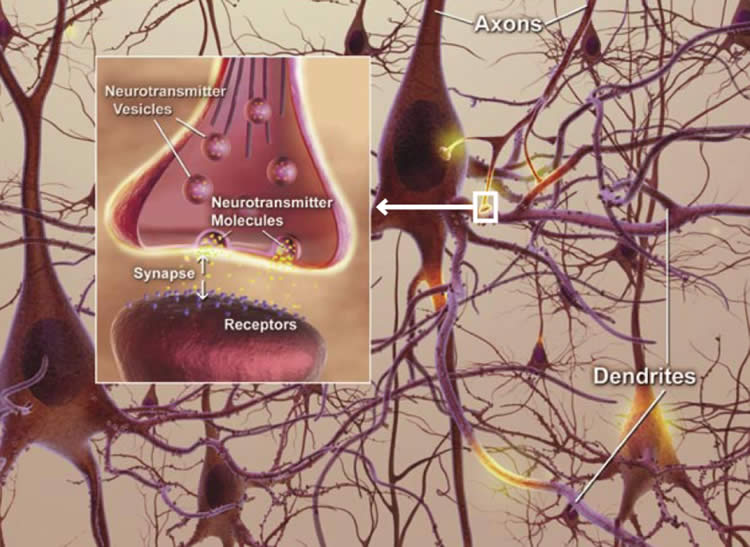
“The interpretation was that about half the spines in the neocortex are long-term repositories for memories while others retain malleability for new memories or forgetting,” Schnitzer said.
Deep and dense
If the same line of thinking held true for the hippocampus as it did for the neocortex, spines in the hipocampus should turn over roughly every 30 days along with the memories they hold. Verifying that idea had been challenging, however, because the hippocampus is deeply buried in the brain and the spines in that region are so densely packed that multiple spines can appear to merge into one.
Schnitzer said there were three components to his team’s ability to track spines in the hippocampus. The first was a technique he reported in 2011 that allows scientists to stably image a single neuron in a living mouse over long time periods. The next was an optical needle, called a microendoscope, that provides high-resolution images of structures deep within the brain.
Even with a stable and high-resolution way of imaging neurons in the hippocampus over time, the team still faced the challenge of distinguishing when spines are gained and lost if they couldn’t tell the difference between a single spine and several merged bulges. “The ability to resolve spines in the hippocampus is right on the hairy edge of our technological capability,” Schnitzer said.
The team overcame that problem with a mathematical model that took into account the limitations of the optical resolution and how that would affect the image datasets depicting the appearances and disappearances of spines.
What Schnitzer and his team found in this analysis is that the region of the hippocampus that stores episodic memories contains spines that all turn over every three to six weeks – roughly the duration of episodic memory in mice.
Schnitzer said that the work confirmed a long-held idea about how the brain stores memories. Using the same techniques, scientists can now probe additional aspects of how memories are formed, remembered and eventually lost at the level of the individual connections between neurons.
Source: Amy Adams – Stanford
Image Credit: Image is credited to the National Institute on Aging and is in the public domain
Original Research: Abstract for “Impermanence of dendritic spines in live adult CA1 hippocampus” by Alessio Attardo, James E. Fitzgerald and Mark J. Schnitzer in Nature. Published online June 22 2015 doi:10.1038/nature14467
Abstract
Impermanence of dendritic spines in live adult CA1 hippocampus
The mammalian hippocampus is crucial for episodic memory formation1 and transiently retains information for about 3–4 weeks in adult mice and longer in humans2. Although neuroscientists widely believe that neural synapses are elemental sites of information storage3, there has been no direct evidence that hippocampal synapses persist for time intervals commensurate with the duration of hippocampal-dependent memory. Here we tested the prediction that the lifetimes of hippocampal synapses match the longevity of hippocampal memory. By using time-lapse two-photon microendoscopy4 in the CA1 hippocampal area of live mice, we monitored the turnover dynamics of the pyramidal neurons’ basal dendritic spines, postsynaptic structures whose turnover dynamics are thought to reflect those of excitatory synaptic connections5, 6. Strikingly, CA1 spine turnover dynamics differed sharply from those seen previously in the neocortex7, 8, 9. Mathematical modelling revealed that the data best matched kinetic models with a single population of spines with a mean lifetime of approximately 1–2 weeks. This implies ~100% turnover in ~2–3 times this interval, a near full erasure of the synaptic connectivity pattern. Although N-methyl-D-aspartate (NMDA) receptor blockade stabilizes spines in the neocortex10, 11, in CA1 it transiently increased the rate of spine loss and thus lowered spine density. These results reveal that adult neocortical and hippocampal pyramidal neurons have divergent patterns of spine regulation and quantitatively support the idea that the transience of hippocampal-dependent memory directly reflects the turnover dynamics of hippocampal synapses.
“Impermanence of dendritic spines in live adult CA1 hippocampus” by Alessio Attardo, James E. Fitzgerald and Mark J. Schnitzer in Nature. Published online June 22 2015 doi:10.1038/nature14467






