Summary: Skin cells may hold the key to explaining why women are more prone to developing autoimmune diseases, such as lupus than men. Researchers found women have more VGLL3 in their skin cells than men. VGLL3 pushes the immune system into overdrive, resulting in the ‘self-attacking’ autoimmune response, the mouse study revealed. Findings strongly implicate VGLL3 as a pivotal catalyst in sex-based autoimmunity.
Source: University of Michigan
It’s one of the great mysteries of medicine, and one that affects the lives of millions of people: Why do women’s immune systems gang up on them far more than men’s do, causing nine times more women to develop autoimmune diseases such as lupus?
Part of the answer, it turns out, may lie in the skin.
New evidence points to a key role for a molecular switch called VGLL3. Three years ago, a team of University of Michigan researchers showed that women have more VGLL3 in their skin cells than men.
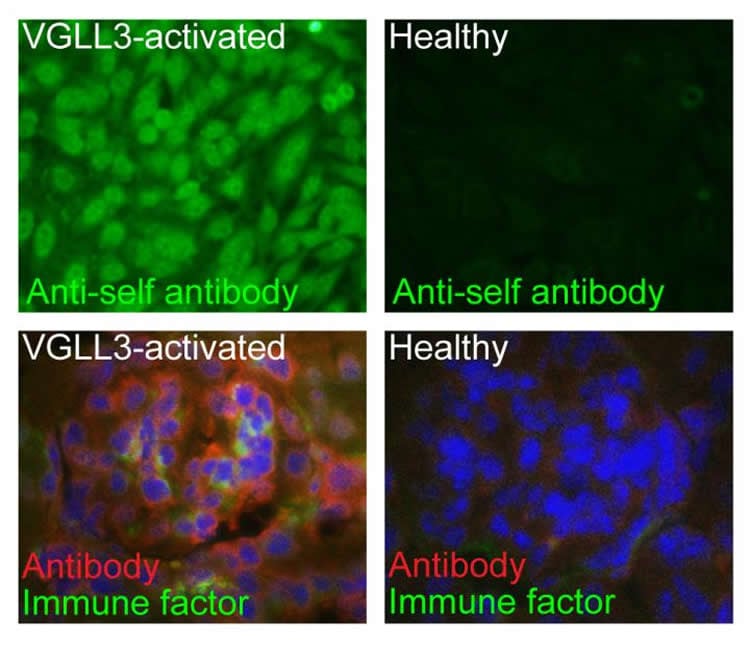
Now, working in mice, they’ve discovered that having too much VGLL3 in skin cells pushes the immune system into overdrive, leading to a “self-attacking” autoimmune response. Surprisingly, this response extends beyond the skin, attacking internal organs too.
Writing in JCI Insight, the team describes how VGLL3 appears to set off a series of events in the skin that trigger the immune system to come running – even when there’s nothing to defend against.
“VGLL3 appears to regulate immune response genes that have been implicated as important to autoimmune diseases that are more common in women, but that doesn’t appear to be regulated by sex hormones,” says Johann Gudjonsson, M.D., Ph.D., who led the research team and is a professor of dermatology at the U-M Medical School.
“Now, we have shown that over-expression of VGLL3 in the skin of transgenic mice is by itself sufficient to drive a phenotype that has striking similarities to systemic lupus erythematosus, including skin rash, and kidney injury.”
Effects of excess VGLL3
Gudjonsson worked with co-first authors Allison Billi, M.D., Ph.D., and Mehrnaz Gharaee-Kermani, Ph.D., and colleagues from several U-M departments, to trace VGLL3’s effects.
They found that extra VGLL3 in skin cells changed expression levels of a number of genes important to the immune system. Expression of many of the same genes is altered in autoimmune diseases like lupus.
The gene expression changes caused by excess VGLL3 wreaked havoc in the mice. Their skin becomes scaly and raw. Immune cells abound, filling the skin and lymph nodes. The mice also produce antibodies against their own tissues, including the same antibodies that can destroy the kidneys of lupus patients.
The researchers don’t yet know what causes female skin cells to have more VGLL3, to begin with. It may be that over evolutionary time females have developed stronger immune systems to fight off infections – but at the cost of increased risk for autoimmune disease if the body mistakes itself for an invader.
The researchers also don’t know what triggers might set off extra VGLL3 activity. But they do know that in men with lupus, the same VGLL3 pathway seen in women with lupus is activated.
Many of the current therapies for lupus, like steroids, come with unwanted side effects, from increased infection risk to cancer. Finding the key factors downstream of VGLL3 may identify targets for new, and potentially safer, therapies that could benefit patients of both sexes.
Lupus, which affects 1.5 million Americans, can cause debilitating symptoms, and current broad-based treatment with steroids can make patients far more vulnerable to infections and cancer.
Patients’ role in future research
Their colleague and senior coauthor Michelle Kahlenberg, M.D., of the U-M Division of Rheumatology, is now recruiting patients with lupus for a study sponsored by U-M’s A. Alfred Taubman Medical Research Institute that could provide answers to these questions and more.
Billi, a resident in dermatology, notes that when she speaks with patients who come to Michigan Medicine’s dermatology clinics for the treatment of the skin problems lupus can cause, she has to acknowledge the limits of current treatment. Even so, she says, patients are eager to take part in studies by contributing skin and DNA samples that could lead to new discoveries about their condition.
“Many patients are frustrated that they’ve had to try multiple therapies, and still nothing is working well,” she says. “To be able to tell them that we’re working on a mouse that has the same disease as them and that we need their help brings out their motivation and interest in research. They know that it’s a long game, and they’re in for it.”
In addition to Gudjonsson, Billi and Kahlenberg, the research team included U-M researchers Mehrnaz Gharaee-Kermani, a co-first author, Joseph Fullmer, Lam C. Tsoi, Brett D. Hill, Xianying Xing, Shannon Estadt, Sonya J. Wolf, Syed Monem Rizvi, Celine C. Berthier, Jeffrey B. Hodgin, Maria A. Beamer, Mrinal K. Sarkar, Yun Liang, Ranjitha Uppala, Shuai Shao, Chang Zeng, Paul W. Harms, Monique E. Verhaegen, John J. Voorhees, Fei Wen, and Andrzej A. Dlugosz, and colleagues from Case Western Reserve University Dennis Gruszka, Jessica Ludwig and Nicole L. Ward.
Funding: The research was funded by the National Institutes of Health and the University of Michigan, with additional support from the Rheumatology Research Foundation, the Dermatology Foundation, the Arthritis National Research Foundation. and the National Psoriasis Foundation. Gudjonsson and Kahlenberg are supported by and are Emerging Scholars of the Taubman Institute. The University has applied for a patent on the VGLL3 mouse model.
Source:
University of Michigan
Media Contacts:
Kara Gavin – University of Michigan
Image Source:
The image is credited to University of Michigan.
Original Research: Open access.
“The female-biased factor VGLL3 drives cutaneous and systemic autoimmunity”
Johann Gudjonsson et al. JCI Insight. doi:10.1172/jci.insight.127291
Abstract
The female-biased factor VGLL3 drives cutaneous and systemic autoimmunity
Autoimmune disease is 4 times more common in women than men. This bias is largely unexplained. Female skin is “autoimmunity prone,” showing upregulation of many proinflammatory genes, even in healthy women. We previously identified VGLL3 as a putative transcription cofactor enriched in female skin. Here, we demonstrate that skin-directed overexpression of murine VGLL3 causes a severe lupus-like rash and systemic autoimmune disease that involves B cell expansion, autoantibody production, immune complex deposition, and end-organ damage. Excess epidermal VGLL3 drives a proinflammatory gene expression program that overlaps with both female skin and cutaneous lupus. This includes increased B cell–activating factor (BAFF), the only current biologic target in systemic lupus erythematosus (SLE); IFN-κ, a key inflammatory mediator in cutaneous lupus; and CXCL13, a biomarker of early-onset SLE and renal involvement. Our results demonstrate that skin-targeted overexpression of the female-biased factor VGLL3 is sufficient to drive cutaneous and systemic autoimmune disease that is strikingly similar to SLE. This work strongly implicates VGLL3 as a pivotal orchestrator of sex-biased autoimmunity.






