Summary: Researchers have developed a new imaging agent, which uses PET scans to target receptors neurons implicated in memory and learning.
Source: SNMMI.
Researchers have developed a new imaging agent that could help guide and assess treatments for people with various neurological diseases, including Alzheimer’s, Parkinson’s, and multiple sclerosis. The agent, which is used in positron emission tomography (PET) scans, targets receptors in nerve cells in the brain that are involved in learning and memory. The study is featured in the April issue of The Journal of Nuclear Medicine.
Swiss and German scientists developed the new PET radioligand, 11C-Me-NB1, for imaging GluN1/GluN2B-containing N-methyl-D-aspartate (NMDA) receptors (a class of glutamate receptor) in nerve cells. When NMDA receptors are activated, there is an increase of calcium (Ca2+) in the cells, but Ca2+ levels that are too high can cause cell death. Medications that block NMDA receptors are therefore used for the treatment of a wide range of neurological conditions from depression, neuropathic pain and schizophrenia to ischemic stroke and diseases causing dementia.
“The significance of the work lies in the fact that we have for the first time developed a useful PET radioligand that can be applied to image the GluN2B receptor subunit of the NMDA receptor complex in humans,” explains Simon M. Ametamey, PhD, of the Institute of Pharmaceutical Sciences, ETH Zurich, in Switzerland. “The availability of such a PET radioligand would not only help to better understand the role of NMDA receptors in the pathophysiology of the many brain diseases in which the NMDA receptor is implicated, but it would also help to select appropriate doses of clinically relevant GluN2B receptor candidate drugs. Administering the right dose of the drugs to patients will help minimize side-effects and lead to improvement in the efficacy of the drugs.”
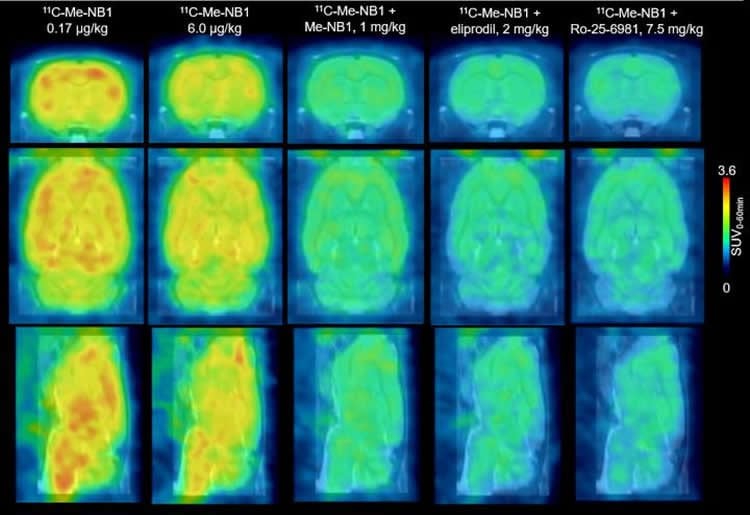
For the study, 11C-Me-NB1 was used in live rats to investigate the dose and effectiveness of eliprodil, a drug that blocks the NMDA GluN2B receptor. PET scans with the new radioligand successfully showed that the receptors are fully occupied at neuroprotective doses of eliprodil. The new radioligand also provided imaging of receptor crosstalk between Sigma-1 receptors, which modulate calcium signaling, and GluN2B-containing NMDA receptors.
Ametamey points out, “These results mean that a new radiopharmaceutical tool is now available for studying brain disorders such as Alzheimer`s disease, Parkinson`s disease and multiple sclerosis, among others. It joins the list of existing PET radiopharmaceuticals used in imaging studies to investigate and understand underlying causes of these brain disorders.” He adds, “Furthermore, future imaging studies using this new radioligand would throw more light on the involvement of NMDA receptors, specifically the GluN2B receptors, in normal physiological processes such as learning and memory, as well as accelerate the development of GluN2B candidate drugs currently under development.”
Funding: This study was financially supported by the Swiss National Science Foundation (Grant Number 160403).
Source: Laurie Callahan – SNMMI
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to SD Krämer et al., ETH Zurich, Zurich, Switzerland.
Original Research: Abstract for “Evaluation of 11C-Me-NB1 as a Potential PET Radioligand for Measuring GluN2B-Containing NMDA Receptors, Drug Occupancy, and Receptor Cross Talk” by Stefanie D. Krämer, Thomas Betzel, Linjing Mu, Ahmed Haider, Adrienne Müller Herde, Anna K. Boninsegni, Claudia Keller, Marina Szermerski, Roger Schibli, Bernhard Wünsch and Simon M. Ametamey in Journal of Nuclear Medicine. Published April 1 2018.
doi:10.2967/jnumed.117.200451
[cbtabs][cbtab title=”MLA”]SNMMI “Novel PET Imaging Agent Could Help Guide Therapy for Brain Diseases.” NeuroscienceNews. NeuroscienceNews, 1 April 2018.
<https://neurosciencenews.com/pet-brain-diseases-8720/>.[/cbtab][cbtab title=”APA”]SNMMI (2018, April 1). Novel PET Imaging Agent Could Help Guide Therapy for Brain Diseases. NeuroscienceNews. Retrieved April 1, 2018 from https://neurosciencenews.com/pet-brain-diseases-8720/[/cbtab][cbtab title=”Chicago”]SNMMI “Novel PET Imaging Agent Could Help Guide Therapy for Brain Diseases.” https://neurosciencenews.com/pet-brain-diseases-8720/ (accessed April 1, 2018).[/cbtab][/cbtabs]
Abstract
Evaluation of 11C-Me-NB1 as a Potential PET Radioligand for Measuring GluN2B-Containing NMDA Receptors, Drug Occupancy, and Receptor Cross Talk
Clinical and preclinical research with modulators at the N-methyl-D-aspartate (NMDA) receptor GluN2B N-terminal domain (NTD) aims for the treatment of various neurologic diseases. The interpretation of the results is hampered by the lack of a suitable NMDA PET tracer for assessing the receptor occupancy of potential drugs. We have developed 11C-Me-NB1 as a PET tracer for imaging GluN1/GluN2B-containing NMDA receptors and used it to investigate in rats the dose-dependent receptor occupancy of eliprodil, a GluN2B NTD modulator. Methods: 11C-Me-NB1 was synthesized and characterized by in vitro displacement binding experiments with rat brain membranes, in vitro autoradiography, and blocking and displacement experiments by PET and PET kinetic modeling. Receptor occupancy by eliprodil was studied by PET with 11C-Me-NB1. Results: 11C-Me-NB1 was synthesized at 290 ± 90 GBq/μmol molar activity, 7.4 ± 1.9 GBq total activity at the end of synthesis (n = 17), and more than 99% radiochemical purity. 11C-Me-NB1 binding in rat brain was blocked in vitro and in vivo by the NTD modulators Ro-25-6981 and eliprodil. Half-maximal receptor occupancy by eliprodil occurred at 1.5 μg/kg. At 1 mg/kg of eliprodil, a dose with reported neuroprotective effects, more than 99.5% of binding sites were occupied. In vitro, 11C-Me-NB1 binding was independent of the σ-1 receptor (Sigma1R), and the Sigma1R agonist (+)-pentazocine did not compete for high-affinity binding. In vivo, a 2.5 mg/kg dose of (+)-pentazocine abolished 11C-Me-NB1–specific binding, indicating an indirect effect of Sigma1R on 11C-Me-NB1 binding. Conclusion: 11C-Me-NB1 is suitable for the in vivo imaging of NMDA GluN1/GluN2B receptors and the assessment of receptor occupancy by NTD modulators. GluN1/GluN2B NMDA receptors are fully occupied at neuroprotective doses of eliprodil. Furthermore, 11C-Me-NB1 enables imaging of GluN1/GluN2B NMDA receptor cross talk.






