Summary: A small molecule, 6-thio-dG, may provide a new approach to treating currently untreatable pediatric brain cancers, researchers report.
Source: Cincinnati Children’s Hospital.
Researchers used an experimental molecular therapy in preclinical laboratory tests to effectively treat several types of deadly pediatric brain cancer and now propose advancing the treatment to clinical testing in children.
Scientists at the Cincinnati Children’s Cancer and Blood Diseases Institute report in the journal Molecular Cancer Therapeutics testing the small molecule 6-thio-2’deoxyguanosine (6-thio-dG) in brain cancer stem cells derived from tumor cells donated by patients. Researchers also tested the treatment in humanized mouse models of pediatric brain cancer.
The cancers include intrinsic pontine glioma (DIPG), high-grade glioma (HGG) and high-risk medulloblastoma (MB).
In preclinical laboratory tests, the targeted molecular therapy crossed the body’s protective blood-brain barrier in mouse models–causing extensive DNA damage in cancer cells and stopping or slowing two untreatable types of pediatric brain cancer. The therapy did not affect non-cancerous cells in the animals, according to the scientists. The therapeutic benefits of 6-thio-dG continued in the animals after treatment was ended.
“These findings show that 6-thio-dG is a promising novel approach to treat therapy resistant pediatric brain tumors and they provide a rationale for clinical testing of this treatment in children with brain tumors,” said Rachid Drissi, PhD, a senior study investigator in the Division of Oncology.
More Research Needed
Although 6-thio-dG does not appear to enter non-cancerous cells, Drissi stressed more research is needed to ensure the compound’s safety before clinical testing in patients. These studies are underway.
Despite decades of research effort and technological advancements, DIPG and other high-risk pediatric brain cancers have continued to defy effective treatment.
This study shows that after injection, 6-thio-dG worked by first crossing the blood-brain barrier of DIPG mouse models–a significant advancement that eliminates a major hurdle to treating brain cancers with drugs. The blood-brain barrier protects the organ from pathogens passing through the circulatory system, making it pharmacologically impenetrable.
Interrupting Immortality
The compound works by triggering extensive DNA damage in brain cancer cells and cancer stem cells. To accomplish this, the compound disrupts the function of telomeres on the chromosomes of brain cancer cells, according to the researchers.
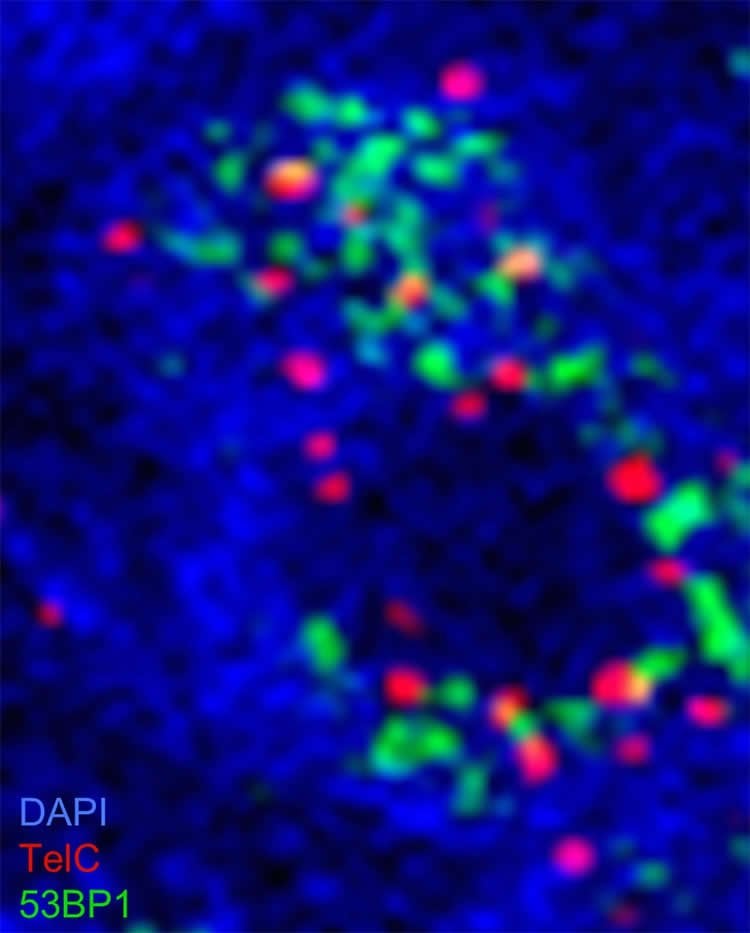
Telomeres are repeated sequences of DNA at the ends of chromosomes that prevent or minimize genetic instability in cells. As cells age, telomeres normally shorten and genetic instability helps kill off the aging cells. But brain cancer cells become immortal because their telomere length continue to be maintained by the enzyme telomerase, making the cells difficult or impossible to kill.
Drissi and his colleagues have shown in earlier studies that 73 percent of DIPG tumors and 50 percent of high-grade gliomas have the enzyme telomerase. Essentially, telomerase helps cancer cells maintain the length of their telomeres.
This led the research team to use a treatment protocol with 6-thio-dG, which already is being tested in preclinical research on lung cancer at another institution. The compound 6-thio-dG is manufactured to be like a similar, naturally occurring ingredient in telomeres. After injection and entrance to the brain, 6-thio-dG is incorporated in cancer cell chromosomes, which causes telomerase-dependent telomere damage.
Collaborators include physicians and scientists from the Brain Tumor Center (Division of Oncology) and Division of Pathology and Laboratory Medicine at Cincinnati Children’s and the Department of Cell Biology, University of Texas Southwestern Medical Center, Dallas.
Funding: Funding support came in part from: CancerFree KIDS; Pediatric Cancer Research Alliance; the Cincinnati Children’s Division of Oncology and Brain Tumor Center.
Source: Nick Miller – Cincinnati Children’s Hospital
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Cincinnati Children’s.
Original Research: Abstract for “Induced Telomere Damage to Treat Telomerase Expressing Therapy-Resistant Pediatric Brain Tumors” by Satarupa Sengupta, Matthew Sobo, Kyungwoo Lee, Shiva Senthil Kumar, Angela R. White, Ilgen Mender, Christine Fuller, Lionel ML Chow, Maryam Fouladi, Jerry W Shay and Rachid Drissi in Molecular Cancer Therapeutics. Published April 17 2018.
doi:10.1158/1535-7163.MCT-17-0792
[cbtabs][cbtab title=”MLA”]Cincinnati Children’s Hospital “Possible Novel Method for Stopping Untreatable Pediatric Brain Cancers.” NeuroscienceNews. NeuroscienceNews, 17 April 2018.
<https://neurosciencenews.com/pediatric-brain-cancer-8824/>.[/cbtab][cbtab title=”APA”]Cincinnati Children’s Hospital (2018, April 17). Possible Novel Method for Stopping Untreatable Pediatric Brain Cancers. NeuroscienceNews. Retrieved April 17, 2018 from https://neurosciencenews.com/pediatric-brain-cancer-8824/[/cbtab][cbtab title=”Chicago”]Cincinnati Children’s Hospital “Possible Novel Method for Stopping Untreatable Pediatric Brain Cancers.” https://neurosciencenews.com/pediatric-brain-cancer-8824/ (accessed April 17, 2018).[/cbtab][/cbtabs]
Abstract
Induced Telomere Damage to Treat Telomerase Expressing Therapy-Resistant Pediatric Brain Tumors
Brain tumors remain the leading cause of cancer-related deaths in children and often are associated with long-term sequelae among survivors of current therapies. Hence, there is an urgent need to identify actionable targets and to develop more effective therapies. Telomerase and telomeres play important roles in cancer, representing attractive therapeutic targets to treat children with poor-prognosis brain tumors such as diffuse intrinsic pontine glioma (DIPG), high-grade glioma (HGG) and high-risk medulloblastoma (MB). We have previously shown that DIPG, HGG and MB frequently express telomerase activity. Here we show that the telomerase-dependent incorporation of 6-thio-2’deoxyguanosine (6-thio-dG), a telomerase substrate precursor analog, into telomeres leads to telomere dysfunction-induced foci (TIFs) along with extensive genomic DNA damage, cell growth inhibition and cell death of primary stem-like cells derived from patients with DIPG, HGG and MB. Importantly, the effect of 6-thio-dG is persistent even after drug withdrawal. Treatment with 6-thio-dG elicits a sequential activation of ATR and ATM pathways, and induces G2/M arrest. In vivo, treatment of mice bearing MB xenografts with 6-thio-dG delays tumor growth, increases in-tumor TIFs and apoptosis. Furthermore, 6-thio-dG crosses the blood-brain barrier and specifically targets tumor cells in an orthotopic mouse model of DIPG. Together, our findings suggest that 6-thio-dG is a promising novel approach to treat therapy-resistant telomerase-positive pediatric brain tumors.






