Findings may have implications for treating compulsive behavior associated with psychiatric disease and eating disorders.
What started as an experiment to probe brain circuits involved in compulsive behavior has revealed a surprising connection with obesity.
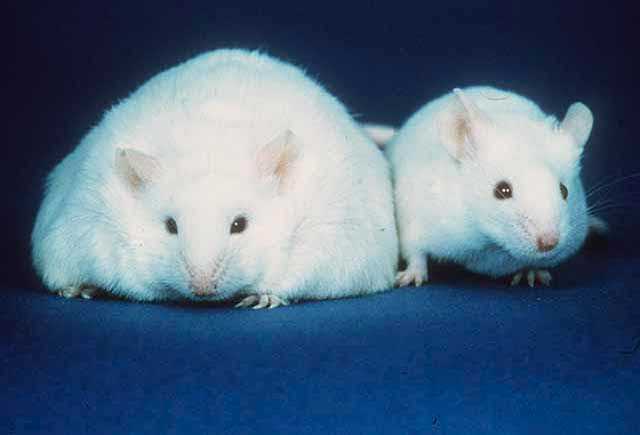
The University of Iowa-led researchers bred mice missing a gene known to cause obesity, and suspected to also be involved in compulsive behavior, with a genetic mouse model of compulsive grooming. The unexpected result was offspring that were neither compulsive groomers nor obese.
The study, published the week of June 10 in the online early edition of the Proceedings of the National Academy of Sciences (PNAS), suggests that the brain circuits that control obsessive-compulsive behavior are intertwined with circuits that control food intake and body weight. The findings have implications for treating compulsive behavior, which is associated with many forms of psychiatric disease, including obsessive-compulsive disorder (OCD), Tourette syndrome, and eating disorders.

UI neuro-psychiatrists Michael Lutter, M.D., Ph.D. and Andrew Pieper, M.D., Ph.D. led the study. The team also included researchers from Stanford University School of Medicine, University of Texas Southwestern Medical Center, Beth Israel Deaconess Medical Center, and Harvard Medical School.
Lutter, an assistant professor of psychiatry, and Pieper, an associate professor of psychiatry and neurology at the UI Carver College of Medicine, both recently arrived at the UI and use mouse models in their laboratories to study human disorders and conditions.
Pieper is interested in compulsive behavior. His mouse model of compulsivity lacks a brain protein called SAPAP3. These mice groom themselves excessively to the point of lesioning their skin, and their compulsive behavior can be effectively treated by fluoxetine, a drug that is commonly used to treat OCD in people.
Lutter works with a mouse that genetically mimics an inherited form of human obesity. This mouse lacks a brain protein known a MC4R. Mutations in the MC4R gene are the most common single-gene cause of morbid obesity and over-eating in people.
“I study MC4R signaling pathways and their involvement in the development of obesity,” Lutter explains. “I’m also interested in how these same molecules affect mood and anxiety and reward, because it’s known that there is a connection between depression and anxiety and development of obesity.”
An old study hinted that in addition to its role in food intake and obesity, MC4R might also play a role in compulsive behavior, which got Lutter and Pieper thinking of ways to test the possible interaction.
“We knew in one mouse you could stimulate excessive grooming through this MC4R pathway and in another mouse a different pathway (SAPAP3) caused compulsive grooming,” Lutter says. “So, we decided to breed the two mice together to see if it would have an effect on compulsive grooming.”
The experiment proved their original hypothesis—knocking out the MC4R protein in the OCD mouse normalized grooming behavior in the animals. In addition, chemically blocking MC4R in the OCD mice also eliminated compulsive grooming. The rescued behavior is mirrored by normalization of a particular pattern of brain cell communication linked to compulsive behavior.
However, the breeding experiment revealed another totally unexpected result. Loss of the SAPAP3 protein from the mice that were obese due to lack of MC4R produced mice of normal weight.
“We had this other, completely shocking finding—we completely rescued body weight and food intake in the double null mouse,” Lutter says. “So, not only were we affecting the brain regions involved in grooming and behavior, but we also affected the brain regions involved in food intake and body weight.”
Although obesity and obsessive-compulsive behavior may seem unrelated, Lutter suggests that the connection may be rooted in the evolutionary need to eat safe, clean food in times of a food abundance, and to lessen this drive when food is scarce.
“Food safety has been an issue through the entire course of human evolution—refrigeration is a relatively recent invention,” he says. “Obsessive behavior, or fear of contamination, may be an evolutionary protection against eating rotten food.”
Oils and fats have lots of calories and nutrients but they also spoil much more easily than less nutrient- and calorie-dense foods like potatoes, onions, or apples.
“I think this circuit that we have uncovered is probably involved in determining whether or not people should eat calorically dense foods,” he says.
Lutter suggests that slight perturbations in this system might lead, on one hand, to disorders that link anxiety and obsessive behavior to limited food selection or intake, such as anorexia nervosa, Tourette syndrome, or OCD, and on the other hand, to obesity, where people over-consume high-fat foods and may have decreased obsessive behavior and anxiety.
“The next step will be to determine how these two pathways communicate with one another, in hopes of identifying new ways to develop drugs to treat either of these disorders,” says Pieper.
Notes about this neuroscience and OCD research
The research was funded by grants from The Hartwell Foundation, the Brain and Behavior Foundation, and the National Institutes of Health DK081185-01, DK081182-01, MH084058-01A1, RO1DK075632, P30DK046200, P30DK057521, F3DK078478). Lutter also was funded by a NARSAD Young Investigator Award.
Written by Jennifer Brown
Contact: Jennifer Brown – University of Iowa
Source: University of Iowa press release
Image Source: The mice image is credited to Oak Ridge National Laboratory and is in the public domain.
Original Research: Full open access research (PDF) for “Double deletion of melanocortin 4 receptors and SAPAP3 corrects compulsive behavior and obesity in mice” by Pin Xu, Brad A. Grueter, Jeremiah K. Britt, Latisha McDaniel, Paula J. Huntington, Rachel Hodge, Stephanie Tran, Brittany L. Mason, Charlotte Lee, Linh Vong, Bradford B. Lowell, Robert C. Malenka, Michael Lutter, and Andrew A. Pieper in PNAS. Published online June 10 2013 doi:10.1073/pnas.1308195110