Summary: Researchers have developed a protein sensor which allows for the observation of nicotine’s movement in cells.
Source: CalTech.
When a person takes a puff on a cigarette, nicotine floods into the brain, latching onto receptors on the surface of neurons and producing feelings of happiness. But nicotine does not simply stay on the surface of cells–the drug actually permeates into neural cells and alters them from the inside out. Now, a team of scientists has developed a protein sensor that glows in the presence of nicotine, allowing the researchers to observe nicotine’s movements in cells and reveal more about the nature of nicotine addiction.
The work was led by Henry Lester, professor of biology at Caltech and previously a visiting scientist at the Janelia Research Campus of the Howard Hughes Medical Institute (HHMI). A paper describing the research appears online on February 4 in the Journal of General Physiology. Lester is also an affiliated faculty member of the Tianqiao and Chrissy Chen Institute for Neuroscience at Caltech.
The endoplasmic reticulum is the equivalent of a cell’s factory and warehouse–the place where proteins are synthesized and packaged in order to be shipped to various other locations both inside and outside of the cell. Nicotinic receptors (nAChRs) are among these proteins; after being manufactured in the endoplasmic reticulum, they then travel to the cell’s surface. When nicotine molecules enter the body, they travel through the bloodstream and reach brain cells, where they meet the nAChRs on the surface of these cells. This triggers the cells’ processes of releasing chemicals of reward and happiness.
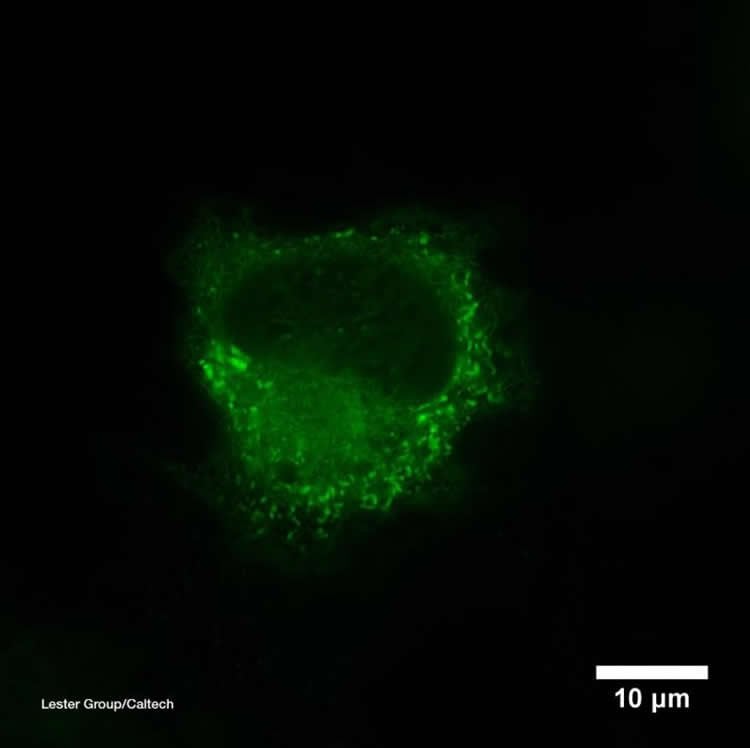
What happens once nicotine has moved into the cells, however, has not been well understood. Lester and others previously found that some nAChRs remain in the “warehouse”–the endoplasmic reticulum–where they, too, can bind to nicotine. Hoping to gain insights into nicotine’s effects within cells, Lester and his team developed a tool called a biosensor to visualize where the drug collects inside of cells. The biosensor is composed of a special protein that can open and close, like a Venus flytrap, and an inactivated fluorescent protein. The sensor is designed to close around nicotine, and this then activates the fluorescent protein to glow brightly, indicating where the nicotine molecules are located and how many are present.
Scientists can put the biosensors into particular parts of a cell–in this work, they placed them in the endoplasmic reticulum and on cells’ surfaces–and watch them light up as nicotine floods in.
By making movies of cells containing biosensors in a lab dish, the team has discovered that nicotine enters into the endoplasmic reticulum within a few seconds of appearing outside a cell. Furthermore, the nicotine levels are more than enough to affect nAChRs during their assembly and to chaperone additional nAChRs on their journey to the cell surface. As a result, the neurons are more sensitive to the nicotine, which enhances the rewarding feelings after a puff on a tobacco cigarette or an e-cigarette. In other words, the more a person smokes, the more quickly and easily the smoker gets a nicotine buzz. This is part of nicotine addiction.
While the movies now focus on isolated neurons in the lab, the scientists want to determine whether nicotine’s intracellular movements are similar in the neurons of live mice. Additionally, they are developing biosensors for other drugs, such as opioids and antidepressants, to observe how these compounds interact inside and outside of cells.
Funding: Funding was provided by the National Institutes of Health, the California Tobacco-Related Disease Research Program, the California Institute for Regenerative Medicine, the Brain & Behavior Research Foundation, HHMI, the Della Martin Foundation, Louis and Janet Fletcher, and Caltech Summer Undergraduate Research Fellowship donors.
Source: Lori Dajose – CalTech
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Caltech/Lester laboratory.
Video Source: Video credited to CalTech.
Original Research: Abstract for “Determining the pharmacokinetics of nicotinic drugs in the endoplasmic reticulum using biosensors” by Amol V. Shivange, Philip M. Borden,Anand K. Muthusamy, Aaron L. Nichols, Kallol Bera, Huan Bao, Ishak Bishara, Janice Jeon, Matthew J. Mulcahy, Bruce Cohen, Saidhbhe L. O’Riordan, Charlene Kim, Dennis A. Dougherty, Edwin R. Chapman, Jonathan Marvin, Loren Looger, and Henry A. Lester in Journal of General Physiology. Published February 4 2019.
doi:10.1085/jgp.201812201
[cbtabs][cbtab title=”MLA”]CalTech”This Is a Neuron on Nicotine.” NeuroscienceNews. NeuroscienceNews, 7 February 2019.
<https://neurosciencenews.com/nicotine-neuron-addiction-10705/>.[/cbtab][cbtab title=”APA”]CalTech(2019, February 7). This Is a Neuron on Nicotine. NeuroscienceNews. Retrieved February 7, 2019 from https://neurosciencenews.com/nicotine-neuron-addiction-10705/[/cbtab][cbtab title=”Chicago”]CalTech”This Is a Neuron on Nicotine.” https://neurosciencenews.com/nicotine-neuron-addiction-10705/ (accessed February 7, 2019).[/cbtab][/cbtabs]
Abstract
Determining the pharmacokinetics of nicotinic drugs in the endoplasmic reticulum using biosensors
Nicotine dependence is thought to arise in part because nicotine permeates into the endoplasmic reticulum (ER), where it binds to nicotinic receptors (nAChRs) and begins an “inside-out” pathway that leads to up-regulation of nAChRs on the plasma membrane. However, the dynamics of nicotine entry into the ER are unquantified. Here, we develop a family of genetically encoded fluorescent biosensors for nicotine, termed iNicSnFRs. The iNicSnFRs are fusions between two proteins: a circularly permutated GFP and a periplasmic choline-/betaine-binding protein engineered to bind nicotine. The biosensors iNicSnFR3a and iNicSnFR3b respond to nicotine by increasing fluorescence at [nicotine] <1 µM, the concentration in the plasma and cerebrospinal fluid of a smoker. We target iNicSnFR3 biosensors either to the plasma membrane or to the ER and measure nicotine kinetics in HeLa, SH-SY5Y, N2a, and HEK293 cell lines, as well as mouse hippocampal neurons and human stem cell–derived dopaminergic neurons. In all cell types, we find that nicotine equilibrates in the ER within 10 s (possibly within 1 s) of extracellular application and leaves as rapidly after removal from the extracellular solution. The [nicotine] in the ER is within twofold of the extracellular value. We use these data to run combined pharmacokinetic and pharmacodynamic simulations of human smoking. In the ER, the inside-out pathway begins when nicotine becomes a stabilizing pharmacological chaperone for some nAChR subtypes, even at concentrations as low as ∼10 nM. Such concentrations would persist during the 12 h of a typical smoker’s day, continually activating the inside-out pathway by >75%. Reducing nicotine intake by 10-fold decreases activation to ∼20%. iNicSnFR3a and iNicSnFR3b also sense the smoking cessation drug varenicline, revealing that varenicline also permeates into the ER within seconds. Our iNicSnFRs enable optical subcellular pharmacokinetics for nicotine and varenicline during an early event in the inside-out pathway.






