Summary: A 20-minute session of non-invasive brain stimulation over four days helps to improve both working and long-term memory in those aged 65 and older.
Source: Boston University
According to the Alzheimer’s Association, an estimated 6.5 million Americans aged 65 and older are living with Alzheimer’s in 2022. That figure is predicted to nearly double by 2050.
Dr. Robert Reinhart, an assistant professor at Boston University’s College of Arts and Sciences and director of the Cognitive & Clinical Neuroscience Laboratory, recognizes the severity of this issue and is working to create treatments to help people with brain disorders.
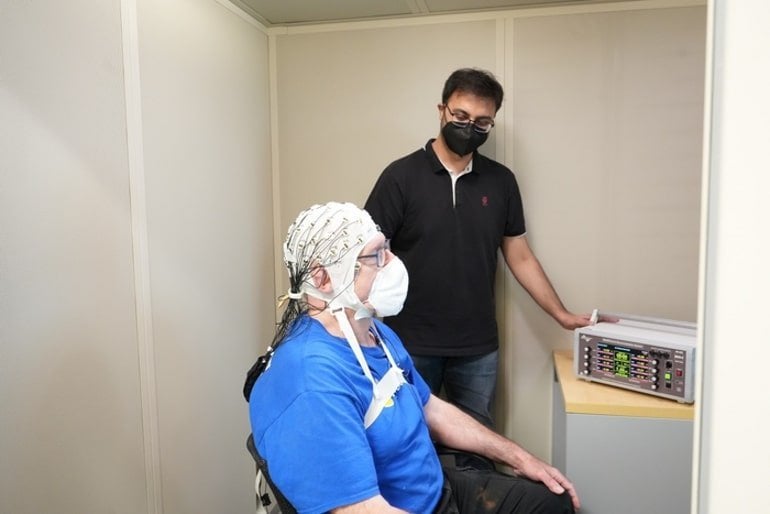
In a recent study published in Nature Neuroscience, Reinhart and his team of researchers explain how their noninvasive treatment delivered through electrodes in a wearable cap can improve memory function in older adults and gets us one step closer to providing a more efficient treatment for memory loss.
Robert Reinhart explains his research, findings, and potential impact in the Q&A below.
What did your study find?
We delivered our noninvasive treatment to target memory function through scalp electrodes and found that electrical brain stimulation for 20 minutes on four consecutive days can improve working memory and long-term memory in individuals 65 years and older for at least one month.
Low-frequency electrode activity improved working memory on day three and day four and one month after intervention, on the other hand, high-frequency improved long-term memory on days two–four and one month after intervention. Our findings demonstrate that the plasticity of the aging brain can be selectively and sustainably altered using these two treatments.
How does this paper expand on your previous study with electrostimulation treatment for memory loss?
In this new study, we used multiple, consecutive days of stimulation for 20 minutes to cause long-lasting memory improvements that lasted one month. Previously, the effects lasted only 50 minutes.
Another important difference is that in this recent study we developed two brain stimulation protocols — one for selectively improving short-term memory via low-frequency parietal stimulation, and another protocol for selectively improving long-term memory via high-frequency prefrontal stimulation.
What are the real-world implications of these findings?
An increasingly older population leads to additional personal, social, healthcare and economic costs. A factor greatly contributing to these costs is the impairment in basic memory systems essential for activities of everyday life, such as making financial decisions or understanding language.
Memory decline varies in severity across individuals during aging, with a rapid decline potentially predicting Alzheimer’s disease and other dementias.
Existing therapeutic approaches for impaired cognition are limited by mixed treatment outcomes, slow improvement, and accompanying risks and side effects. For those reasons, there’s an urgent need to develop innovative therapeutic interventions that can provide rapid and sustainable improvements with minimal side effects.
Clinically, this is important because there are people with only short-term memory problems and others with only long-term memory problems. So, having tools in hand that can address each of these memory systems is of great value.
What’s next?
Further research is needed to determine whether these effects can last beyond one month and whether these specific methods can also enhance memory function in individuals with impaired cognition due to brain disorders and in those at risk for dementia.
About this neurotech and memory research news
Author: Thalia Plata
Source: Boston University
Contact: Thalia Plata – Boston University
Image: The image is credited to Robert Reinhart
Original Research: Closed access.
“Long-lasting, dissociable improvements in working memory and long-term memory in older adults with repetitive neuromodulation” by Robert Reinhart et al. Nature Neuroscience
Abstract
Long-lasting, dissociable improvements in working memory and long-term memory in older adults with repetitive neuromodulation
The development of technologies to protect or enhance memory in older people is an enduring goal of translational medicine. Here we describe repetitive (4-day) transcranial alternating current stimulation (tACS) protocols for the selective, sustainable enhancement of auditory–verbal working memory and long-term memory in 65–88-year-old people.
Modulation of synchronous low-frequency, but not high-frequency, activity in parietal cortex preferentially improved working memory on day 3 and day 4 and 1 month after intervention, whereas modulation of synchronous high-frequency, but not low-frequency, activity in prefrontal cortex preferentially improved long-term memory on days 2–4 and 1 month after intervention.
The rate of memory improvements over 4 days predicted the size of memory benefits 1 month later. Individuals with lower baseline cognitive function experienced larger, more enduring memory improvements.
Our findings demonstrate that the plasticity of the aging brain can be selectively and sustainably exploited using repetitive and highly focalized neuromodulation grounded in spatiospectral parameters of memory-specific cortical circuitry.







