Summary: Researchers report bioelectrical potential is a driving force for stem cells to generate different neuron types during embryogenesis.
Source: University of Geneva.
The cerebral cortex is a highly developed brain region, which allows intellectual functions such as conscious perception, anticipation of events and language. These functions are mediated by specific sets of neuronal circuits. To understand how these circuits emerge during development, researchers from the University of Geneva (UNIGE), Switzerland, in collaboration with an american team, investigated what enables neuronal stem cells to generate successive subtypes of neurons as the embryo grows. By measuring the electrical activity of these progenitors, they found that akin to a battery getting charged, membrane voltage values increase as the embryo develops and new neurons are being created. To test the role of this electrical charge, neuroscientists experimentally manipulated progenitor voltage values, which allowed them to select which type of neuron was being born. These results, published in Cell, reveal an unexpected role for bioelectric cell properties in the generation of neuronal diversity.
The cerebral cortex is characterized by a diversity of neuronal cell type which assemble during development to form circuits, underlying functions such as skilled movements and sensory integration. These circuits emerge in two stages: first during embryogenesis, when neurons are born, and later on after we are born, when neurons contact each other to form circuits that are chiseled by the environment.
In the embryo, different types of cortical neurons are sequentially created in a well-defined order by stem cells called neuronal progenitors, located deep within the brain. “We wondered how these progenitors were able to know what type of neurons to make at each embryonic age,” explains Denis Jabaudon, professor in the Department of Basic Neurosciences at the Faculty of Medicine of the UNIGE.
Electricity drives progenitor behaviour
To address this question, the UNIGE team took an unusual approach. “Usually genes are considered as the stars of cellular development, explains Ilaria Vitali, a researcher in the team. Here, we focused on another player, the electrical properties of these progenitors.” While the role of electrical properties in the normal workings of neuronal circuits is well established, there was until now only little information on a role for electrical charge in progenitor properties. Using a pipette smaller than two microns in diameter to pierce the cell membrane, the neuroscientists were able to measure the electrical charge of neuronal progenitors as the cortex was forming in mouse embryos. “What we found is that as the embryo grows and the types of neurons generate become more complex, progenitor voltage values increased,” says Denis Jabaudon.
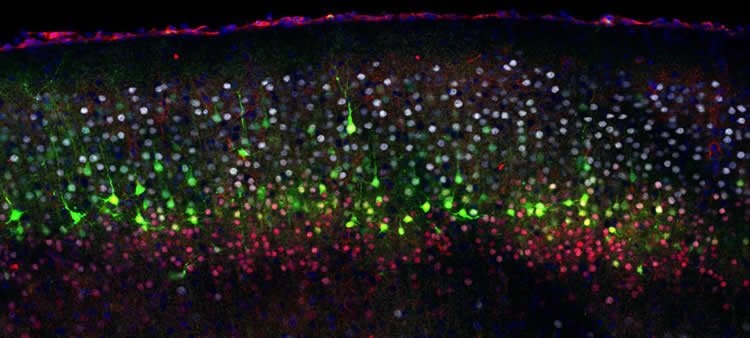
An electrical switch to generate distinct neuronal subtypes
To manipulate this process, the scientists next genetically modified embryonic cells. “We expressed small synthetic channels at the surface of the progenitors, which allowed us to manipulate their voltage at will by charging or discharging these cells,” explains Sabine Fièvre, a co-author in the study and researcher in the UNIGE team. “And it turns out that if the progenitors are artificially charged early in embryogenesis, they prematurely generate normally later-born neurons.” “Conversely, if we discharge the progenitors, they generate neurons that are normally born earlier in the embryo’s life,” adds Denis Jabaudon.
These results demonstrate that the bioelectric activity of progenitors plays a central role in generating neuronal diversity. This could help explain how some neurological disorders associated with abnormal electrical activity, such as epilepsy, affect brain development during embryogenesis.
Source: Denis Jabaudon – University of Geneva
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to UNIGE.
Original Research: Abstract for “Progenitor Hyperpolarization Regulates the Sequential Generation of Neuronal Subtypes in the Developing Neocortex” by Ilaria Vitali, Sabine Fièvre, Ludovic Telley, Polina Oberst, Sebastiano Bariselli6, Laura Frangeul, Natalia Baumann, John J. McMahon, Esther Klingler, Riccardo Bocchi, Jozsef Z. Kiss, Camilla Bellone, Debra L. Silver, and Denis Jabaudon in Cell. Published July 26 2018.
doi:10.1016/j.cell.2018.06.036
[cbtabs][cbtab title=”MLA”]University of Geneva”Electricity Sparks Neural Diversity During Brain Development.” NeuroscienceNews. NeuroscienceNews, 26 July 2018.
<https://neurosciencenews.com/neural-diversity-brain-development-9613/>.[/cbtab][cbtab title=”APA”]University of Geneva(2018, July 26). Electricity Sparks Neural Diversity During Brain Development. NeuroscienceNews. Retrieved July 26, 2018 from https://neurosciencenews.com/neural-diversity-brain-development-9613/[/cbtab][cbtab title=”Chicago”]University of Geneva”Electricity Sparks Neural Diversity During Brain Development.” https://neurosciencenews.com/neural-diversity-brain-development-9613/ (accessed July 26, 2018).[/cbtab][/cbtabs]
Abstract
Progenitor Hyperpolarization Regulates the Sequential Generation of Neuronal Subtypes in the Developing Neocortex
Highlights
•Apical progenitor (AP) resting membrane potential decreases during corticogenesis
•AP hyperpolarization allows progression from direct to indirect neurogenesis
•AP hyperpolarization drives neuronal diversity via repression of Wnt signaling
Summary
During corticogenesis, ventricular zone progenitors sequentially generate distinct subtypes of neurons, accounting for the diversity of neocortical cells and the circuits they form. While activity-dependent processes are critical for the differentiation and circuit assembly of postmitotic neurons, how bioelectrical processes affect nonexcitable cells, such as progenitors, remains largely unknown. Here, we reveal that, in the developing mouse neocortex, ventricular zone progenitors become more hyperpolarized as they generate successive subtypes of neurons. Experimental in vivo hyperpolarization shifted the transcriptional programs and division modes of these progenitors to a later developmental status, with precocious generation of intermediate progenitors and a forward shift in the laminar, molecular, morphological, and circuit features of their neuronal progeny. These effects occurred through inhibition of the Wnt-beta-catenin signaling pathway by hyperpolarization. Thus, during corticogenesis, bioelectric membrane properties are permissive for specific molecular pathways to coordinate the temporal progression of progenitor developmental programs and thus neocortical neuron diversity.






