Summary: Study reveals the molecular mechanism that allows neural networks to grow and branch out.
Source: Yale
Our nervous system is composed of billions of neurons that speak to one another through their axons and dendrites. When the human brain develops, these structures branch out in a beautifully intricate yet poorly understood way that allows nerve cells to form connections and send messages throughout the body. And now, Yale researchers have discovered the molecular mechanism behind the growth of this complex system.
Their findings are published in Science Advances.
“Neurons are highly branched cells, and they’re like this because each neuron makes a connection with thousands of other neurons,” says Joe Howard, Ph.D., Eugene Higgins Professor of Molecular Biophysics and Biochemistry and professor of physics, and senior researcher of the study.
“We’re working on this branching process—how do branches form and grow? That is what’s underlying the whole way the nervous system works.”
The team studied neuronal growth in fruit flies as they matured from embryos into larvae. To visualize this process, they tagged neurons with fluorescent markers and imaged them on a spinning disk microscope. Because neurons reside just under the cuticle [outermost layer], the researchers were able to observe this process in real time in live larvae.
After imaging the neurons at different stages of development, the team was able to create time-lapse movies of the growth.
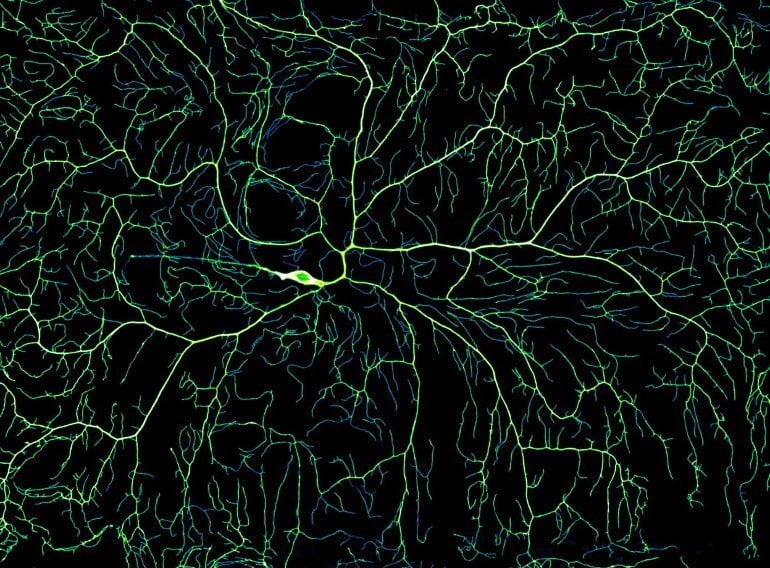
In the earliest stages of development, the sensory neurons began with only two or three dendrites. But in as little as five days, they blossomed into big, tree-like structures with thousands of branches.
Analysis of dendritic tips revealed their dynamic and stochastic (randomly determined) growth, which fluctuated among growing, shrinking, and paused states.
“Before our study, there was a theory that neurons may be dilating and deflating like a balloon,” says Sonal Shree, Ph.D., associate research scientist and lead author of the study. “And we found that no, they’re not inflating like a balloon, but rather growing and branching their tips.”
“We found that we can completely explain neuronal growth and the overall morphology in terms of just what the tips of the cells are doing,” says Sabyasachi Sutradhar, Ph.D., associate research scientist and joint lead author of the study.
“This means that now we can focus on the tips, because if we can understand how they work, then we can understand how the whole shape of the cell comes about,” says Howard.
There is a whole world of branching in biology, from the veins and arteries of the circulatory system to the bronchioles of the lung. Howard’s lab hopes that better understanding of branching at the cellular level will also shed light on these processes at the molecular and tissue levels.
About this neuroscience research news
Author: Isabella Backman
Source: Yale
Contact: Isabella Backman – Yale
Image: The image is credited to Howard Lab
Original Research: Open access.
“Dynamic instability of dendrite tips generates the highly branched morphologies of sensory neurons” by Sonal Shree et al. Science Advances
Abstract
Dynamic instability of dendrite tips generates the highly branched morphologies of sensory neurons
The highly ramified arbors of neuronal dendrites provide the substrate for the high connectivity and computational power of the brain. Altered dendritic morphology is associated with neuronal diseases.
Many molecules have been shown to play crucial roles in shaping and maintaining dendrite morphology. However, the underlying principles by which molecular interactions generate branched morphologies are not understood.
To elucidate these principles, we visualized the growth of dendrites throughout larval development of Drosophila sensory neurons and found that the tips of dendrites undergo dynamic instability, transitioning rapidly and stochastically between growing, shrinking, and paused states.
By incorporating these measured dynamics into an agent-based computational model, we showed that the complex and highly variable dendritic morphologies of these cells are a consequence of the stochastic dynamics of their dendrite tips.
These principles may generalize to branching of other neuronal cell types, as well as to branching at the subcellular and tissue levels.