Researchers at the Babraham Institute and University of Massachusetts Medical School in the United States have developed a new model to study motor neuron degeneration and have used this to identify three genes involved in the neurodegeneration process. These findings could have relevance for understanding the progression of amyotrophic lateral sclerosis (ALS) and other forms of motor neuron disease (MND). ALS is the most common form of adult-onset motor neuron disease and kills over 1,200 people a year in the UK.
The researchers developed a new model to study neurodegeneration in the common fruit fly (Drosophila melanogaster). In contrast to other methods used to study neurodegeneration such as looking at changes in eye morphology or studying larval stages, the researchers focused their attention on studying the neurons in a fruit fly’s leg. Using the leg allows the detailed study of a single motor neuron, the nerve cell involved in passing signals from the brain to a muscle. Compared to studying larval stages, the model also permits genuine ageing studies as changes in neurons can be observed in flies of different ages. The adult fruit fly can live for over two months in the lab. Furthermore, the fruit fly also provides the benefits of rapid development (ten days from egg to adult), allowing high-throughput genetic screens.
As described online in Current Biology, the researchers used the new model to study the role of a protein central to the development of ALS called TDP-43. Specifically overexpressing TDP-43 in fly legs caused neurodegeneration. Exposing these flies to a mutagen and looking for offspring showing reduced neurodegeneration allowed the researchers to identify three genes implicated in mediating the effects of TDP-43. One, shaggy/GSK3, was already know to be associated with the neurodegeneration process but two of them, hat-trick and xmas-2, were new discoveries.
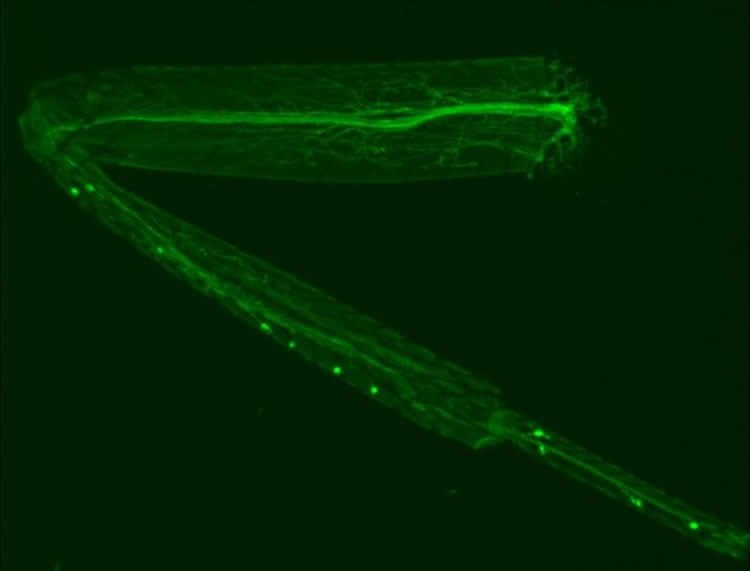
Dr. Jemeen Sreedharan, Senior Research Fellow in the signalling research programme at the Babraham Institute and lead author on the paper said: “We’re extremely excited about our new approach to using the power of Drosophila genetics. Never before has anyone been able to study adult neurodegeneration in an invertebrate system with such ease. By modelling the earliest stages of ALS (synaptic and axon degeneration) we have identified three intriguing genetic suppressors of degeneration in the fly and are now validating these results in mammalian neuronal cultures. We hope that by using the fly we can accelerate progress towards eventually developing therapies for ALS and other neurodegenerative diseases.”
FundingL Funding support for this research was provided by the Medical Research Council, a Motor Neurone Disease Association Lady Edith Wolfson Fellowship and the Max Rosenfeld Fund. The Babraham Institute receives strategic funding (a total of £28.8M in 2013-14) from the Biotechnology and Biological Sciences Research Council (BBSRC).
Source: Louisa Wood – Babraham Institute
Image Source: The image is credited to Babraham Institute
Original Research: Abstract for “Age-Dependent TDP-43-Mediated Motor Neuron Degeneration Requires GSK3, hat-trick, and xmas-2” by Jemeen Sreedharan, Lukas J. Neukomm, Robert H. Brown Jr., and Marc R. Freeman in Current Biology. Published online July 30 2015 doi:10.1016/j.cub.2015.06.045
Abstract
Age-Dependent TDP-43-Mediated Motor Neuron Degeneration Requires GSK3, hat-trick, and xmas-2
Highlights
•Mosaic analysis in the fly leg is a powerful tool to study neurodegeneration
•TDP-43 causes age-dependent dying-back degeneration of adult motor neurons
•Loss of GSK3, hat-trick, or xmas-2 suppresses TDP-43 toxicity
•TDP-43-mediated axon degeneration is distinct from Wallerian degeneration
Summary
The RNA-processing protein TDP-43 is central to the pathogenesis of amyotrophic lateral sclerosis (ALS), the most common adult-onset motor neuron (MN) disease [ 1–4 ]. TDP-43 is conserved in Drosophila, where it has been the topic of considerable study, but how TDP-43 mutations lead to age-dependent neurodegeneration is unclear and most approaches have not directly examined changes in MN morphology with age [ 5 ]. We used a mosaic approach to study age-dependent MN loss in the adult fly leg where it is possible to resolve single motor axons, NMJs and active zones, and perform rapid forward genetic screens. We show that expression of TDP-43Q331K caused dying-back of NMJs and axons, which could not be suppressed by mutations that block Wallerian degeneration. We report the identification of three genes that suppress TDP-43 toxicity, including shaggy/GSK3, a known modifier of neurodegeneration [ 6 ]. The two additional novel suppressors, hat-trick and xmas-2, function in chromatin modeling and RNA export, two processes recently implicated in human ALS [ 7, 8 ]. Loss of shaggy/GSK3, hat-trick, or xmas-2 does not suppress Wallerian degeneration, arguing TDP-43Q331K-induced and Wallerian degeneration are genetically distinct processes. In addition to delineating genetic factors that modify TDP-43 toxicity, these results establish the Drosophila adult leg as a valuable new tool for the in vivo study of adult MN phenotypes.
“Age-Dependent TDP-43-Mediated Motor Neuron Degeneration Requires GSK3, hat-trick, and xmas-2” by Jemeen Sreedharan, Lukas J. Neukomm, Robert H. Brown Jr., and Marc R. Freeman in Current Biology. Published online July 30 2015 doi:10.1016/j.cub.2015.06.045






