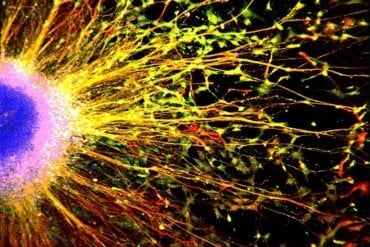
Summary: According to researchers, intestinal bacteria can accelerate Alzheimer’s development.
Source: Lund University.
New research from Lund University in Sweden has shown that intestinal bacteria can accelerate the development of Alzheimer’s disease. According to the researchers behind the study, the results open up the door to new opportunities for preventing and treating the disease
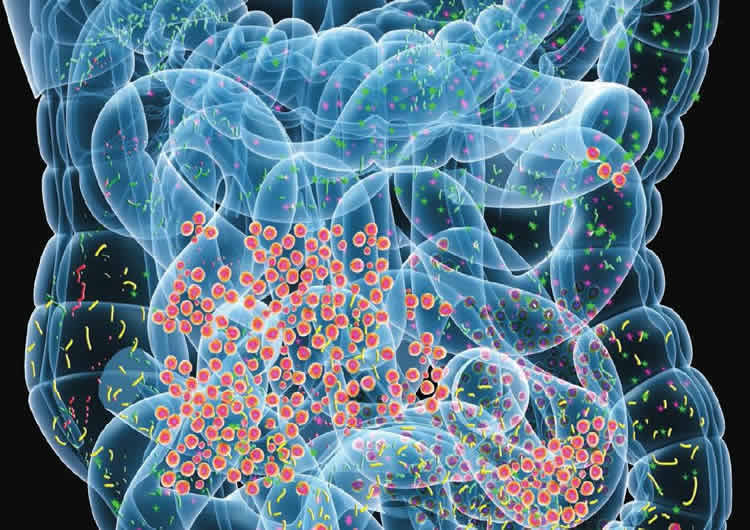
Because our gut bacteria have a major impact on how we feel through the interaction between the immune system, the intestinal mucosa and our diet, the composition of the gut microbiota is of great interest to research on diseases such as Alzheimer’s. Exactly how our gut microbiota composition is composed depends on which bacteria we receive at birth, our genes and our diet.
By studying both healthy and diseased mice, the researchers found that mice suffering from Alzheimer’s have a different composition of gut bacteria compared to mice that are healthy. The researchers also studied Alzheimer’s disease in mice that completely lacked bacteria to further test the relationship between intestinal bacteria and the disease. Mice without bacteria had a significantly smaller amount of beta-amyloid plaque in the brain. Beta-amyloid plaques are the lumps that form at the nerve fibres in cases of Alzheimer’s disease.
To clarify the link between intestinal flora and the occurrence of the disease, the researchers transferred intestinal bacteria from diseased mice to germ-free mice, and discovered that the mice developed more beta-amyloid plaques in the brain compared to if they had received bacteria from healthy mice.
“Our study is unique as it shows a direct causal link between gut bacteria and Alzheimer’s disease. It was striking that the mice which completely lacked bacteria developed much less plaque in the brain”, says researcher Frida Fåk Hållenius, at the Food for Health Science Centre.
“The results mean that we can now begin researching ways to prevent the disease and delay the onset. We consider this to be a major breakthrough as we used to only be able to give symptom-relieving antiretroviral drugs.”
Funding: The research is a result of an international collaboration between Associate Professor Frida Fåk Hållenius and doctoral student Nittaya Marungruang, both at the Food for Health Science Centre in Lund, and a research group at the Ecole Polytechnique Federale de Lausanne in Switzerland. The collaboration has now expanded to include researchers from Germany and Belgium in connection with receiving a SEK 50 million EU grant.
The researchers will continue to study the role of bacteria in the development of Alzheimer’s disease, and test entirely new types of preventive and therapeutic strategies based on the modulation of the gut microbiota through diet and new types of probiotics.
Source: Frida Fåk Hållenius – Lund University
Image Source: NeuroscienceNews.com image is adapted from Lund University press release.
Original Research: Full open access research for “Reduction of Abeta amyloid pathology in APPPS1 transgenic mice in the absence of gut microbiota” by T. Harach, N. Marungruang, N. Duthilleul, V. Cheatham, K. D. Mc Coy, G. Frisoni, J. J. Neher, F. Fåk, M. Jucker, T. Lasser & T. Bolmont in Scientific Reports. Published online February 8 2017 doi:10.1038/srep41802
[cbtabs][cbtab title=”MLA”]Lund University “Gut Bacteria May Play a Role in Alzheimer’s Disease.” NeuroscienceNews. NeuroscienceNews, 10 February 2017.
<https://neurosciencenews.com/microbiota-alzheimers-6096/>.[/cbtab][cbtab title=”APA”]Lund University (2017, February 10). Gut Bacteria May Play a Role in Alzheimer’s Disease. NeuroscienceNew. Retrieved February 10, 2017 from https://neurosciencenews.com/microbiota-alzheimers-6096/[/cbtab][cbtab title=”Chicago”]Lund University “Gut Bacteria May Play a Role in Alzheimer’s Disease.” https://neurosciencenews.com/microbiota-alzheimers-6096/ (accessed February 10, 2017).[/cbtab][/cbtabs]
Abstract
Reduction of Abeta amyloid pathology in APPPS1 transgenic mice in the absence of gut microbiota
Alzheimer’s disease is the most common form of dementia in the western world, however there is no cure available for this devastating neurodegenerative disorder. Despite clinical and experimental evidence implicating the intestinal microbiota in a number of brain disorders, its impact on Alzheimer’s disease is not known. To this end we sequenced bacterial 16S rRNA from fecal samples of Aβ precursor protein (APP) transgenic mouse model and found a remarkable shift in the gut microbiota as compared to non-transgenic wild-type mice. Subsequently we generated germ-free APP transgenic mice and found a drastic reduction of cerebral Aβ amyloid pathology when compared to control mice with intestinal microbiota. Importantly, colonization of germ-free APP transgenic mice with microbiota from conventionally-raised APP transgenic mice increased cerebral Aβ pathology, while colonization with microbiota from wild-type mice was less effective in increasing cerebral Aβ levels. Our results indicate a microbial involvement in the development of Abeta amyloid pathology, and suggest that microbiota may contribute to the development of neurodegenerative diseases.
“Reduction of Abeta amyloid pathology in APPPS1 transgenic mice in the absence of gut microbiota” by T. Harach, N. Marungruang, N. Duthilleul, V. Cheatham, K. D. Mc Coy, G. Frisoni, J. J. Neher, F. Fåk, M. Jucker, T. Lasser & T. Bolmont in Scientific Reports. Published online February 8 2017 doi:10.1038/srep41802