Summary: Healing a traumatic brain injury (TBI) might actually start in the digestive system. A new study reveals that short-term antibiotic treatment can significantly reduce neuroinflammation and brain cell death following a TBI.
By remodeling the gut microbiome, the treatment flushes out harmful bacteria and allows beneficial species—specifically Parasutterella excrementihominis and Lactobacillus johnsonii—to flourish. These “helper” bacteria appear to regulate peripheral immunity, preventing the gut-brain axis from sending scrambled signals that would otherwise hinder the brain’s ability to repair itself.
Key Facts
- The Antibiotic Effect: Short-term antibiotics after a TBI were found to reduce lesion size and limit neurodegeneration by cleaning the gut of inflammatory bacteria.
- Key Bacterial Players: The study identified Parasutterella excrementihominis and Lactobacillus johnsonii as essential drivers for cellular repair and regulators of inflammation.
- Immune Powerhouse: Given that 70% of the body’s immune system regulation originates in the gut, a balanced microbiome is critical for the brain’s recovery process.
- Scrambled Signals: A TBI disrupts the bidirectional gut-brain axis; if the gut remains out of balance, it creates a feedback loop that makes neuroinflammation worse.
- Long-term Prevention: Researchers believe that breaking this inflammatory cycle in the acute stage could significantly reduce the long-term risk of developing Alzheimer’s, Parkinson’s, or dementia.
Source: Houston Methodist
What if healing the brain after traumatic injury starts in the gut?
In a new study published in Nature Communications Biology, Houston Methodist researchers led by Sonia Villapol, Ph.D., found that short-term antibiotic treatment significantly reduced neuroinflammation and neurodegeneration following traumatic brain injury (TBI) by altering the gut microbiome in animal models.
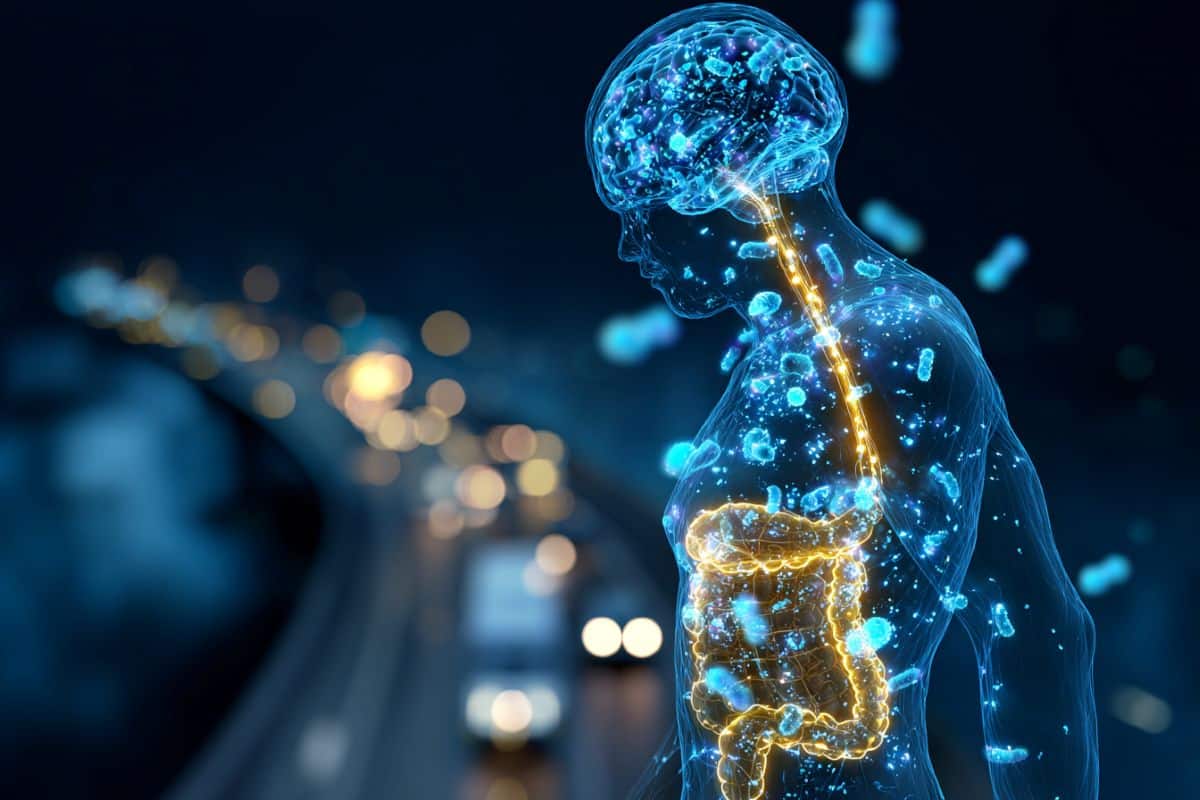
“We found that antibiotic treatment following TBI can reduce harmful gut bacteria, decrease lesion size and limit cell death,” said Villapol, an associate professor in the Department of Neurosurgery at Houston Methodist.
“Our results support a gut–brain mechanism in which microbiome changes influence peripheral immunity and, in turn, neuroinflammation after TBI.¨
Administering antibiotics cleans the gut of harmful bacteria, allowing beneficial bacteria to flourish. The study found that two helpful bacteria, Parasutterella excrementihominis and Lactobacillus johnsonii, are key to driving cell repair. According to Villapol, they could also be major regulators for peripheral inflammation in the body.
Notably, 70% of immune system regulation is generated by the gut microbiome. During gut imbalance, the bidirectional nature of the brain-gut axis can wreak havoc throughout the entire body.
“Our brains are constantly sending signals to the rest of our bodies. Following a traumatic brain event, those signals can get scrambled and disrupt other organs, including our digestive system,” Villapol said. “If the gut stays out of balance, the brain may have a harder time healing.”
There are an estimated 4 million traumatic brain injuries a year in the U.S. Recent studies indicate that TBI-induced gut microbiome imbalance may even contribute to the development of neurodegenerative diseases like Parkinson’s, Alzheimer’s and dementia.
Villapol’s lab is focused on investigating and developing new neuroprotective treatments to fight inflammation linked with neurodegenerative disease. “If we can break neuroinflammation in the acute or chronic stage, we can reduce the risk of developing Alzheimer’s or dementia,” Villapol said.
The next phase of the research will focus on bioengineering Parasutterella excrementihominis and Lactobacillus johnsonii to further develop precision therapies to reduce neuroinflammation.
Other collaborators in the study include Hannah Flinn, Austin Marshall, Morgan Holcomb, Marissa Burke, Goknur Kara, Leonardo Cruz-Pineda, Sirena Soriano and Todd J. Treangen.
Funding: This work was supported in part by grants from Houston Methodist Research Institute and the National Institutes of Health (NIH).
Key Questions Answered:
A: It sounds counterintuitive, but your gut controls about 70% of your immune system. When the brain is injured, it sends “stress signals” to the gut that cause harmful bacteria to take over. Using antibiotics to “reset” the gut allows beneficial bacteria to take the lead, which then sends calming, anti-inflammatory signals back to the brain to help it heal.
A: The study specifically identified two “super-bacteria” (P. excrementihominis and L. johnsonii) that are key to this process. While general probiotics are good, the future of this research is in “precision therapies”—bioengineering these specific bacteria into targeted treatments for TBI patients.
A: There is growing evidence that it can. When a TBI causes long-term gut dysfunction, the resulting chronic neuroinflammation can act as a “slow burn” that contributes to neurodegenerative diseases like Alzheimer’s and Parkinson’s later in life.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neurology research news
Author: Ashley White
Source: Houston Methodist
Contact: Ashley White – Houston Methodist
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Antibiotic-induced gut microbiome remodeling reduces neuroinflammation in traumatic brain injury” by Hannah Flinn, Austin Marshall, Morgan Holcomb, Marissa Burke, Goknur Kara, Leonardo Cruz-Pineda, Sirena Soriano, Todd J. Treangen & Sonia Villapol. Communications Biology
DOI:10.1038/s42003-026-09737-1
Abstract
Antibiotic-induced gut microbiome remodeling reduces neuroinflammation in traumatic brain injury
Traumatic brain injury induces neuroinflammation and gut microbiome dysbiosis, yet the effects of short-term antibiotic treatment on these processes remain poorly understood. To address this, male mice received controlled brain injuries followed by a brief course of oral antibiotics.
Antibiotic treatment reduced bacterial abundance in feces and altered microbial diversity, with more pronounced shifts after two injuries.
Despite this disruption, antibiotic-treated mice exhibited smaller lesion volumes, reduced cell death, attenuated microglial and macrophage activation, lower pro-inflammatory cytokine levels, and decreased astrogliosis and peripheral immune cell infiltration compared with vehicle-treated mice after two injuries.
In the gut, increasing injury severity was associated with villus shortening and loss of mucus-producing cells, and antibiotic treatment further modified these injury-related changes.
Circulating levels of short-chain fatty acids and associated microbial metabolic functions were reduced by antibiotic exposure. In contrast, germ-free mice showed increased lesion volumes and exacerbated gliosis following brain injury.
Long-read metagenomic sequencing identified Parasutterella excrementihominis and Lactobacillus johnsonii as taxa that persisted despite antibiotic treatment.
Collectively, these results suggest that antibiotics can reduce brain damage after injury through mechanisms not explained by short-chain fatty acids, while also highlighting potential drawbacks of altering the gut microbiome.