Summary: Researchers have discovered a new learning rule for a specific type of excitatory synaptic connection in the hippocampus.
Source: IST Austria
IST Austria Professor Peter Jonas and team identify a new learning rule.
“Fire together, wire together” is the famous abridged version of the Hebbian rule. It states that neurons in the brain adapt during the learning process, a mechanism which is called neuronal plasticity. Hebb’s theory dates back to the 1940s and subsequent research in neuroscience has further corroborated it. Today, we also know that different factors play a critical role, such as timing of firing, order of activity, and functional connectivity, as cutting-edge technologies allow examining subcellular processes with extraordinary precision.
Recently, scientists at the Institute of Science and Technology Austria (IST Austria) discovered a new learning rule for a specific type of excitatory synaptic connection in the hippocampus. Their study was now published in the renowned journal Nature Communications on May 13. These synapses are located in the so-called CA3 region of the hippocampus, which plays a critical role for storage and recall of spatial information in the brain. One of its hallmark properties is that memory recall can even be triggered by incomplete cues. This enables the network to complete neuronal activity patterns, a phenomenon termed pattern completion.
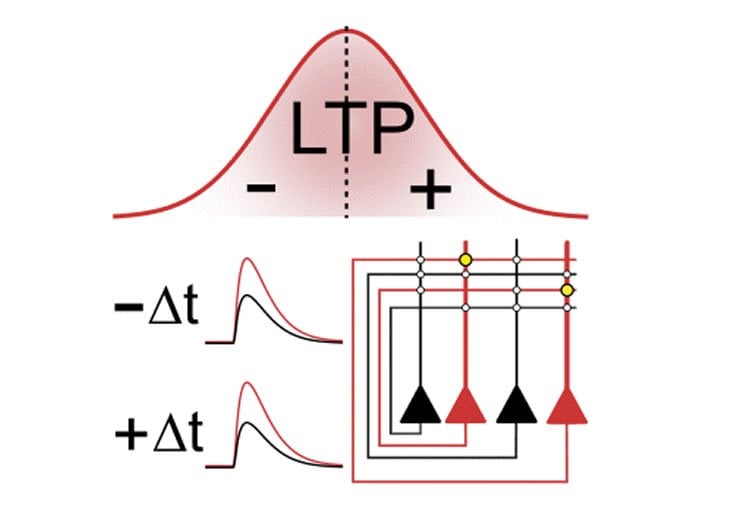
Professor Peter Jonas and his team, including postdoc José Guzmán and PhD student Rajiv Mishra, investigated how the strength of connections between neurons is adjusted, taking into account the relative timing of firing neurons. In neuroscience, this is known as spike-timing-dependent plasticity or STDP. According to the STDP rule, neuron A has to fire just before neuron B so that the synaptic connection becomes stronger with time. In the case of a reverse order—neuron B fires before neuron A—the connection between the neurons may become weaker.
Yet in apparent contrast to this rule, the team of Professor Jonas discovered in their experiments that a reverse order also leads to stronger connections between the investigated synapses (CA3–CA3 recurrent excitatory synapses). Surprisingly, a potentiation takes place independent of the order of firing. So if the sequence is not important at these particular synapses, why is this the case?
To address this question, the authors performed various cutting-edge measurements with extremely high precision. These included patch-clamp recordings to control which neurons fire at what time, imaging of calcium molecules, which play a critical role in synaptic plasticity, and subcellular recordings of electrical signals in dendrites. All data resulted in the same symmetric summation curves. Thus, the unusual induction curve of potentiation is generated by the properties of calcium signaling, which is in turn explained by the characteristics of electrical signaling in dendrites.
The scientists subsequently investigated what happens if a huge number of neurons is being connected via excitatory synapses in a network model. To this end, they ran computer simulations after incorporating different plasticity induction rules. They compared the results of simulations with the new symmetric plasticity induction rule with those of a conventional rule. The outcome clearly demonstrated that patterns could be better restored from partial cues when the new symmetric rule was applied. Professor Jonas: “The new plasticity induction rule may explain why learning in vivo occurs robustly under a variety of behavioral conditions. For example, it may explain storage and recall of cell assembly patterns of freely moving animals in open fields, as previously found by the systems neuroscience groups of IST Austria (O’Neill et al., 2008)”.
The new data seem to be in contrast to classical STDP induction rules at other glutamatergic synapses. Do they violate the Hebb rule? Professor Jonas: “If you read the classical Hebb text carefully, it states: ‘If the axon of a cell A is near enough to excite cell B […], A’s efficiency, as one of the cells firing B, is increased’. However, there is no mentioning of depression. So the new data do not violate Hebb’s postulate, but may confirm it in the literal sense”.
Funding: Supported by the Fond zur Förderung der Wissenschaftlichen Forschung (P 24909-B24) and the European Union (European Research Council Advanced Grant 268548 to P.J.).
Source: IST Austria
Image Source: This NeuroscienceNews.com image is adapted fromt the IST Austria press release.
Original Research: Full open access research for “Symmetric spike timing-dependent plasticity at CA3–CA3 synapses optimizes storage and recall in autoassociative networks” by Rajiv K. Mishra, Sooyun Kim, Segundo J. Guzman and Peter Jonas in Nature Communications. Published online May 13 2016 doi:10.1038/ncomms11552
[cbtabs][cbtab title=”MLA”]IST Austria. “New Learning Rule For Pattern Completion.” NeuroscienceNews. NeuroscienceNews, 13 May 2016.
<https://neurosciencenews.com/learning-synaptic-plasticity-ca3-4240/>.[/cbtab][cbtab title=”APA”]IST Austria. (2016, May 11). New Learning Rule For Pattern Completion. NeuroscienceNews. Retrieved May 13, 2016 from https://neurosciencenews.com/learning-synaptic-plasticity-ca3-4240/[/cbtab][cbtab title=”Chicago”]IST Austria. “New Learning Rule For Pattern Completion.” NeuroscienceNews.
https://neurosciencenews.com/learning-synaptic-plasticity-ca3-4240/ (accessed May 13, 2016).[/cbtab][/cbtabs]
Abstract
Symmetric spike timing-dependent plasticity at CA3–CA3 synapses optimizes storage and recall in autoassociative networks
CA3–CA3 recurrent excitatory synapses are thought to play a key role in memory storage and pattern completion. Whether the plasticity properties of these synapses are consistent with their proposed network functions remains unclear. Here, we examine the properties of spike timing-dependent plasticity (STDP) at CA3–CA3 synapses. Low-frequency pairing of excitatory postsynaptic potentials (EPSPs) and action potentials (APs) induces long-term potentiation (LTP), independent of temporal order. The STDP curve is symmetric and broad (half-width ~150 ms). Consistent with these STDP induction properties, AP–EPSP sequences lead to supralinear summation of spine [Ca2+] transients. Furthermore, afterdepolarizations (ADPs) following APs efficiently propagate into dendrites of CA3 pyramidal neurons, and EPSPs summate with dendritic ADPs. In autoassociative network models, storage and recall are more robust with symmetric than with asymmetric STDP rules. Thus, a specialized STDP induction rule allows reliable storage and recall of information in the hippocampal CA3 network.
“Symmetric spike timing-dependent plasticity at CA3–CA3 synapses optimizes storage and recall in autoassociative networks” by Rajiv K. Mishra, Sooyun Kim, Segundo J. Guzman and Peter Jonas in Nature Communications. Published online May 13 2016 doi:10.1038/ncomms11552






