Summary: The measure of iron in the brain may help doctors predict which patients with Parkinson’s disease are most likely to develop dementia.
Source: UCL
A cutting-edge MRI technique to detect iron deposits in different brain regions can track declines in thinking, memory and movement in people with Parkinson’s disease, finds a new UCL-led study.
The findings, published in the Journal of Neurology, Neurosurgery, and Psychiatry, suggest that measures of brain iron might eventually help predict which people with Parkinson’s will develop dementia.
“Iron in the brain is of growing interest to people researching neurodegenerative diseases such as Parkinson’s and dementias. As you get older, iron accumulates in the brain, but it’s also linked to the build-up of harmful brain proteins, so we’re starting to find evidence that it could be useful in monitoring disease progression, and potentially even in diagnostics,” said the study’s lead author, Dr Rimona Weil (UCL Queen Square Institute of Neurology).
The study involved 97 people with Parkinson’s disease, who had been diagnosed within the last 10 years, along with 37 people without the condition, as a control (comparison) group. They were tested for their thinking and memory as well as for their motor function.
Parkinson’s disease is a progressive condition of brain degeneration resulting in tremors, stiffness and slowness of movement. Close to 50% of people with the condition end up developing dementia, but the timing and severity vary substantially.
Currently there are no reliable measures to track Parkinson’s progression in the brain, so clinicians rely on monitoring symptoms. Conventional brain imaging fails to track progression until quite a late stage, when large-scale brain volume loss can be detected.
Iron accumulates in people’s brains as part of the normal ageing process, partly due to increased permeability in the blood-brain barrier. Excess iron can have toxic effects leading to proteins being irreversibly modified. Recent studies have found that when proteins linked to Alzheimer’s disease (amyloid and tau, which are also linked to Parkinson’s dementia) build up, iron also accumulates in the affected brain areas.
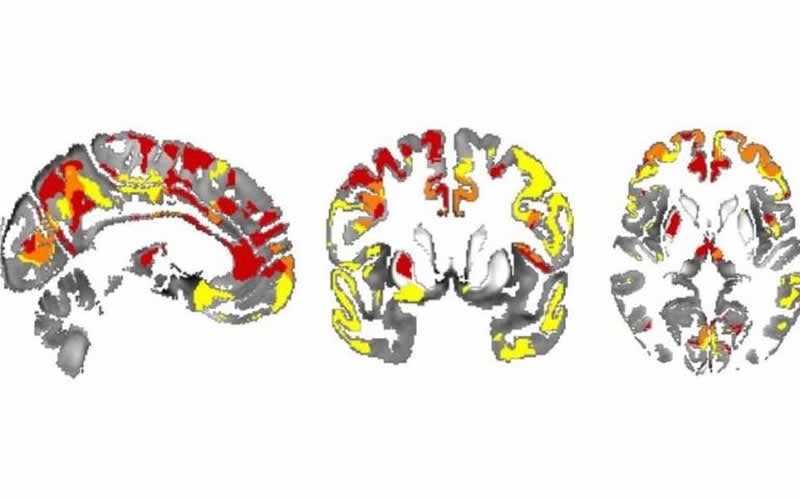
In the current study, researchers used a new technique, called quantitative susceptibility mapping,* to map iron levels in the brain based on MRI (magnetic resonance imaging) scans. They found that iron accumulation in the hippocampus and thalamus brain regions was associated with poor memory and thinking scores. Iron in the putamen brain region was associated with poor movement scores, suggesting a more advanced stage of the disease.
In Parkinson’s disease, the hippocampus and thalamus are known to be associated with thinking and memory, and the putamen with movement scores, so the researchers say it’s very promising that iron deposition was specifically detected in those areas.
The findings suggest that iron deposition could be valuable to track if a treatment is working in a clinical trial, and might eventually be helpful for early diagnosis of Parkinson’s or other neurodegenerative diseases.
Dr Weil has previously found in a 2019 study** that a suite of vision tests may be helpful to predict cognitive decline in Parkinson’s. She and her colleagues hope that further research will determine if the vision tests and iron measures could be helpful to predict which people with dementia are likely to develop dementia.
First author, PhD student George Thomas (UCL Queen Square Institute of Neurology), said: “It’s really promising to see measures like this which can potentially track the varying progression of Parkinson’s disease, as it could help clinicians devise better treatment plans for people based on how their condition manifests.”
Co-author Dr Julio Acosta-Cabronero (Tenoke Ltd. and Wellcome Centre for Human Neuroimaging, UCL) added: “We were surprised at how well the iron levels measured in different regions of the brain with MRI were correlated with cognitive and motor skills. We hope that brain iron measurement could be useful for a wide range of conditions, such as to gauge dementia severity or to see which brain regions are affected by other movement, neuromuscular and neuroinflammatory disorders, stroke, traumatic brain injury and drug abuse.”
The researchers are now following up the same study participants to see how their disease is progressing, whether they develop dementia, and how such measures correlate with changes in iron levels over time.
Funding: The study was supported by Wellcome, the National Institute for Health Research, the Medical Research Council, Parkinson’s UK, Movement Disorders Society, ESRC, and the Cure Parkinson’s Trust.
Source:
UCL
Media Contacts:
Chris Lane – UCL
Image Source:
The image is credited to George Thomas et al.
Original Research: Open access
“Brain iron deposition is linked with cognitive severity in Parkinson’s disease”. George Thomas et al.
Journal of Neurology, Neurosurgery, and Psychiatry doi:10.1136/jnnp-2019-322042.
Abstract
Brain iron deposition is linked with cognitive severity in Parkinson’s disease
Background Dementia is common in Parkinson’s disease (PD) but measures that track cognitive change in PD are lacking. Brain tissue iron accumulates with age and co-localises with pathological proteins linked to PD dementia such as amyloid. We used quantitative susceptibility mapping (QSM) to detect changes related to cognitive change in PD.
Methods We assessed 100 patients with early-stage to mid-stage PD, and 37 age-matched controls using the Montreal Cognitive Assessment (MoCA), a validated clinical algorithm for risk of cognitive decline in PD, measures of visuoperceptual function and the Movement Disorders Society Unified Parkinson’s Disease Rating Scale part 3 (UPDRS-III). We investigated the association between these measures and QSM, an MRI technique sensitive to brain tissue iron content.
Results We found QSM increases (consistent with higher brain tissue iron content) in PD compared with controls in prefrontal cortex and putamen (p<0.05 corrected for multiple comparisons). Whole brain regression analyses within the PD group identified QSM increases covarying: (1) with lower MoCA scores in the hippocampus and thalamus, (2) with poorer visual function and with higher dementia risk scores in parietal, frontal and medial occipital cortices, (3) with higher UPDRS-III scores in the putamen (all p<0.05 corrected for multiple comparisons). In contrast, atrophy, measured using voxel-based morphometry, showed no differences between groups, or in association with clinical measures. Conclusions Brain tissue iron, measured using QSM, can track cognitive involvement in PD. This may be useful to detect signs of early cognitive change to stratify groups for clinical trials and monitor disease progression.