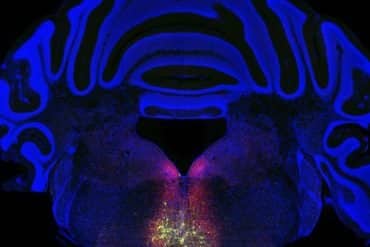
Summary: Developing a human brain is a high-stakes race where timing is everything. For a neuron to function, it must divide, migrate, and mature in a precise sequence. A new review highlights a unique epigenetic mechanism called histone bivalency that acts as the brain’s internal clock.
By placing both “green light” (active) and “red light” (repressive) molecular marks on the same gene simultaneously, the brain keeps critical maturation genes poised like runners at a starting line—ready to go, but held back until the exact moment they are needed.
Key Facts
- The “Bivalent” Switch: Histone bivalency occurs when a gene carries two opposing epigenetic signals. This keeps the gene “poised”—silent but capable of rapid activation.
- The Developmental Clock: If the “red light” is removed too early, neurons mature prematurely, skipping vital migration steps and leading to a malfunctioning brain.
- Adult Brain Surprises: Researchers unexpectedly found hundreds of bivalent genes in mature adult neurons. Traditionally thought to exist only in embryos, these marks persist long after development ends.
- Stress Response & Memory: In adults, these “poised” genes are linked to stress responses and cell death, suggesting bivalency allows mature neurons to react instantly to injury. They may also serve as a “epigenetic memory” of the cell’s developmental history.
Source: Estonian Research Council
The brain is the most complex organ in the human body. Different parts of the brain perform a variety of functions, all of which are necessary for it to operate in one way or another. These functions are carried out by neurons, which communicate with one another in intricate ways.
There are many different subtypes of neurons, which differ from each other in gene expression, cell structure, and function.
For the brain to perform all its functions, the correct number of the right types of neurons must emerge during development. Newborn neurons must migrate from their birthplace to the right location and form connections – synapses – with precisely the right target cells.
Neuronal development proceeds through several stages. Initially, neuronal precursor cells proliferate. They then begin to differentiate – transforming into specific types of neurons – and migrate to their correct position in the brain.
Finally, the cells mature and establish connections with other cells. All of these stages must occur at exactly the right time and in the right order. If maturation happens too early or too late, a neuron cannot form the correct connections, and the brain will not function properly.
The transition of neurons from one developmental stage to the next is governed by epigenetic marks – molecular signals that switch gene expression on and off. Kärt Mätlik, head of the neuroepigenetics research group at TalTech and lead author of the study, compares these marks to traffic lights on the DNA.
“Some marks give a green light (“produce this gene product”) and others a red light (“this gene does not need to be expressed right now”). For example, in a neuronal precursor cell, genes required for cell proliferation during the division stage have a green light.
“In a mature neuron, however, those same division-related genes must be shown a red light, because their activation there can lead to uncontrolled cell proliferation – that is, cancer – or to cell death,” she explained.
Some genes, however, carry both a red and a green light simultaneously. This combination of epigenetic marks is known as histone bivalency. Genes marked by bivalency resemble runners poised at the starting line: ready to move, but waiting for the right moment.
Recently published studies have shown that this mechanism holds back the expression of maturation-related genes in neuronal precursor cells until the time is right. If the repressive signal is artificially removed from bivalent genes in a precursor cell, maturation-related genes are expressed prematurely, the cell matures too early, and skips other critical developmental stages.
These findings have led to the conclusion that the balance between the two signals acts as a built-in clock, ensuring that neuronal maturation only occurs once the preceding developmental stages have been completed.
Beyond this, it has emerged – somewhat unexpectedly – that hundreds of genes remain bivalent even in mature adult brain cells, where developmental processes have long since concluded.
“Histone bivalency has previously been associated primarily with very early development. Surprisingly, however, studies in recent years – including our own earlier work – have shown that some genes remain bivalent in mature neurons,” said Mätlik.
As part of the recent review article, the researchers examined which types of genes are maintained in a bivalent state in the adult brain.
“We found that genes that are bivalent across multiple different neuronal populations are linked to the stress response and cell death. This suggests that bivalency might allow mature neurons to respond rapidly to, for example, stress or injury.
“On the other hand, some bivalent genes in mature neurons were also required during those same cells’ earlier development – meaning bivalency could function as a kind of memory of a cell’s developmental history and the choices made along the way,” she noted.
How the localisation of bivalent marks is regulated, and what their precise functions are in the adult brain, remains to be determined by future research. Kärt Mätlik and her neuroepigenetics research group are continuing to investigate these questions using methods that allow them to track and modify changes in bivalency at different stages of neuronal development, including in human neurons.
The review article by scientists from TalTech and Rockefeller University was published in the journal Genes & Development and summarises current knowledge on histone bivalency in neuronal development.
Key Questions Answered:
A: Think of it like a “pause” button on a high-speed video. The cell needs those maturation genes to be ready to fire instantly, but if they fire too early, the neuron stops moving and gets stuck in the wrong part of the brain. Bivalency keeps the engine running while the car is still in park.
A: If the repressive signal is lost, the neuron matures instantly. It essentially “quits school” before it has learned where to go or how to connect. This can lead to neurodevelopmental disorders or, in some cases, uncontrolled cell growth (cancer) if the division genes aren’t properly turned off.
A: This was the study’s big surprise. In adults, these poised genes seem to be a “break glass in case of emergency” system. If the brain is injured or under extreme stress, these bivalent genes can be activated immediately to help the cell survive or, if necessary, trigger programmed cell death to protect the rest of the network.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this genetics and neurodevelopment research news
Author: Merilin Reede
Source: Estonian Research Council
Contact: Merilin Reede – Estonian Research Council
Image: The image is credited to Neuroscience News
Original Research: Closed access.
“Histone bivalency in CNS development” by Kärt Mätlik, Eve-Ellen Govek, and Mary E. Hatten. Genes & Development
DOI:10.1101/gad.352306.124
Abstract
Histone bivalency in CNS development
Neuronal maturation is guided by changes in the chromatin landscape that control developmental gene expression programs.
Histone bivalency, the co-occurrence of activating and repressive histone modifications, has emerged as an epigenetic feature of developmentally regulated genes during neuronal maturation.
Although initially associated with early embryonic development, recent studies have shown that histone bivalency also exists in differentiated and mature neurons.
In this review, we discuss methods to study bivalency in specific populations of neurons and summarize emerging studies on the function of bivalency in central nervous system neuronal maturation and in adult neurons.