Summary: A new study reveals a defective version of astrocytes may be linked to the build up of alpha synuclein and could spur Parkinson’s disease. The findings show the important role glial cells play in Parkinson’s and offers insights into new targets for therapies to fight the neurodegenerative disease.
Source: Cell Press.
Researchers from the University of Barcelona have shown that defective versions of human brain cells called astrocytes are linked to the buildup of a toxic protein that is the hallmark of Parkinson’s disease. The studied astrocytes, derived from Parkinson’s disease patients with a genetic mutation that affects cell clean-up functions, caused more accumulation of the toxin, alpha-synuclein, than those derived from healthy individuals. The work, which appears January 10 in the journal Stem Cell Reports, suggests an important role for glial cells in Parkinson’s disease and offers potential new targets for developing therapies.
“We have shown that astrocytes play a crucial role in Parkinson’s disease. Our results demonstrate that Parkinson’s disease astrocytes transfer a toxic protein to dopamine-producing neurons,” says co-first author Angelique di Domenico, former postdoctoral fellow at the University of Barcelona’s Bellvitge Biomedical Research Institute.
Star-shaped astrocytes extend branching tendrils around synapses and along blood vessels. One previous postmortem study found that Parkinson’s patients had a buildup of alpha-synuclein–a protein that accumulates to form Lewy bodies, the signature pathology of Parkinson’s disease–in their neurons as well as their astrocytes. It was this observation that led the researchers to examine astrocytes’ role in the disease.
Using cells derived from Parkinson’s patients with an LRRK2 mutation, the researchers generated stem-cell-derived glia cells. Fifteen percent of Parkinson’s cases are tied to an inherited gene mutation, and 7 percent of all cases–inherited and sporadic–are linked to a large protein complex called LRRK2. LRRK2’s main function is still unknown, but it seems to play a role in mitochondrial dynamics and autophagy, a process through which cells break down and rebuild their damaged components.
The researchers then used CRISPR gene editing to track the toxic alpha-synuclein as it was generated by the stem-cell-derived astrocytes and transferred to dopamine-producing neurons.
“We found Parkinson’s disease astrocytes to have fragmented mitochondria, as well as several disrupted cellular degradation pathways, leading to the accumulation of alpha-synuclein,” di Domenico says.
The accumulation of alpha-synuclein caused the targeted neurons’ projecting branches–axons and dendrites–to shorten and disintegrate, resulting in neuronal death, she says.
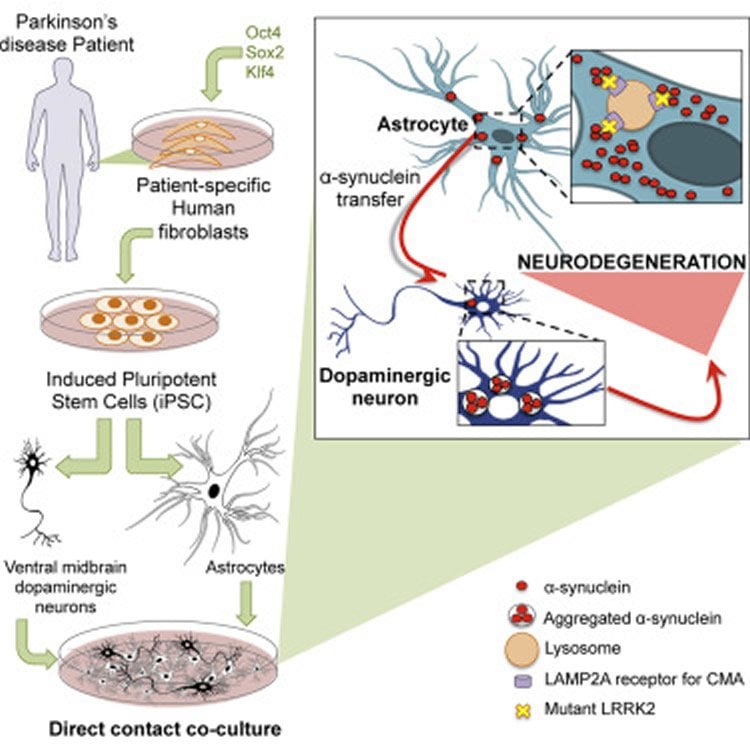
In contrast, when healthy astrocytes were cultured with neurons from Parkinson’s disease patients, axons and dendrites regenerated and alpha-synuclein was prevented from accumulating, ultimately restoring neuronal function, she notes.
The researchers used a drug–developed to treat abnormal intracellular buildup of toxic materials–on the Parkinson’s disease astrocytes. “We were elated to see after treatment that the cellular degradation processes were restored and alpha-synuclein was completely cleared from the Parkinson’s disease astrocytes,” di Domenico says. “These results pave the way to new therapeutic strategies that block pathogenic interactions between neurons and glial cells.”
Next steps involve investigating astrocytes from the 85 to 90 percent of Parkinson’s cases that are sporadic, with no known genetic cause.
Funding: This work was supported by the European Research Council-ERC, the Spanish Ministry of Economy and Competitiveness, Instituto de Salud Carlos III, AGAUR, and CERCA Programme / Generalitat de Catalunya.
Source: Carly Britton – Cell Press
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to di Domenico/Stem Cell Reports.
Original Research: Open access research for “Patient-Specific iPSC-Derived Astrocytes Contribute to Non-Cell-Autonomous Neurodegeneration in Parkinson’s Disease” by Angelique di Domenico, Giulia Carola, Carles Calatayud, Meritxell Pons-Espinal, Juan Pablo Muñoz, Yvonne Richaud-Patin, Irene Fernandez-Carasa, Marta Gut, Armida Faella, Janani Parameswaran, Jordi Soriano, Isidro Ferrer, Eduardo Tolosa, Antonio Zorzano, Ana Maria Cuervo, Angel Raya, and Antonella Consiglio in Stem Cell Reports. Published January 10 2019.
doi:10.1016/j.stemcr.2018.12.011
[cbtabs][cbtab title=”MLA”]Cell Press “Defective Glial Cells Can Push Neurons Toward Parkinson’s Disease.” NeuroscienceNews. NeuroscienceNews, 10 January 2019.
<https://neurosciencenews.com/glial-cells-parkinsons-10494/>.[/cbtab][cbtab title=”APA”]Cell Press(2019, January 10). Defective Glial Cells Can Push Neurons Toward Parkinson’s Disease. NeuroscienceNews. Retrieved January 10, 2019 from https://neurosciencenews.com/glial-cells-parkinsons-10494/[/cbtab][cbtab title=”Chicago”]Cell Press “Defective Glial Cells Can Push Neurons Toward Parkinson’s Disease.” https://neurosciencenews.com/glial-cells-parkinsons-10494/ (accessed January 10, 2019).[/cbtab][/cbtabs]
Abstract
Patient-Specific iPSC-Derived Astrocytes Contribute to Non-Cell-Autonomous Neurodegeneration in Parkinson’s Disease
Parkinson’s disease (PD) is associated with the degeneration of ventral midbrain dopaminergic neurons (vmDAns) and the accumulation of toxic α-synuclein. A non-cell-autonomous contribution, in particular of astrocytes, during PD pathogenesis has been suggested by observational studies, but remains to be experimentally tested. Here, we generated induced pluripotent stem cell-derived astrocytes and neurons from familial mutant LRRK2 G2019S PD patients and healthy individuals. Upon co-culture on top of PD astrocytes, control vmDAns displayed morphological signs of neurodegeneration and abnormal, astrocyte-derived α-synuclein accumulation. Conversely, control astrocytes partially prevented the appearance of disease-related phenotypes in PD vmDAns. We additionally identified dysfunctional chaperone-mediated autophagy (CMA), impaired macroautophagy, and progressive α-synuclein accumulation in PD astrocytes. Finally, chemical enhancement of CMA protected PD astrocytes and vmDAns via the clearance of α-synuclein accumulation. Our findings unveil a crucial non-cell-autonomous contribution of astrocytes during PD pathogenesis, and open the path to exploring novel therapeutic strategies aimed at blocking the pathogenic cross talk between neurons and glial cells.