Summary: Zolgensma, an FDA approved gene therapy, supplements the production of the SMN protein to improve motor neuron function in children with spinal muscular atrophy after just one dose.
Source: Seattle Children’s Hospital
When Arabella Smygov was diagnosed with spinal muscular atrophy (SMA) type 1 at 3 months old, the first recommendation Dr. Fawn Leigh, a neurologist at Seattle Children’s, gave her parents, Sarah and Vitaliy, was to wait on searching for information about SMA online.
This is because up until a few years ago, SMA type 1 was a fatal diagnosis. Most of the information available online painted a bleak picture. Babies diagnosed with SMA type 1, the most severe and common form of the neurodegenerative disease, usually don’t survive beyond age 2 and if they do, they require full support for breathing from a ventilator.
Leigh had good reason for wanting her parents to have hope for Arabella’s future. Two treatments, including the first-ever drug approved for the condition by the U.S. Food and Drug Administration (FDA) in 2016 called Spinraza, and Zolgensma, a gene therapy approved by the drug agency this month, are rapidly changing the trajectory for children with Arabella’s condition.
“I always remember back to when I had to offer my first SMA diagnosis,” Leigh said. “I was heartbroken to tell this young couple that we didn’t have anything for their baby. Now, we’re planning a future for these babies because we have not one, but two good treatment options.”
Family turns to neuromuscular specialist
At Arabella’s 2-month checkup, her pediatrician noticed she wasn’t moving as much as other babies her age. Wanting to get the opinion of a neuromuscular specialist, she referred the family to Leigh in Seattle Children’s Neurosciences Center.
In their first appointment, Leigh observed what she thought were the hallmark symptoms of SMA.
“These babies are generally very floppy and weak like Raggedy Ann dolls,” she said. “They can’t lift their head or arms. Their natural history is very grim. They never sit. They never really roll over and if they do, they lose that ability.”
A blood test confirmed Leigh’s suspicion. Arabella had SMA type 1. She explained to Sarah and Vitaliy how SMA is caused by a mutation in the SMN1 gene, which encodes for a protein that motor neurons — nerve cells that control muscle contraction — need to survive. When that gene is mutated, infants with SMA type 1, continue to lose motor neurons as they encounter stress, injury and illness. Over time, they lose all motor strength and mobility.
“We were desperate to do whatever we could for Arabella, but we completely trusted Dr. Leigh and followed her advice,” Vitaliy said. “We didn’t look it up online. Instead, we took some time to process the information she gave us before scheduling a follow up appointment to talk about our options.”
Gene therapy presented as potential option
In their next appointment, Leigh presented both Spinraza and Zolgensma as options for Arabella.
Zolgensma, she explained was an experimental gene therapy still in clinical trials, but the preliminary data were very promising. Babies were standing, walking and thriving several years after receiving the therapy. These studies also showed the earlier the therapy could be delivered, the better the outcome for the child. Leigh thought Arabella might be a good candidate for the therapy.
“Our first reaction was that we just wanted to help Arabella in the best way possible,” Sarah said. “My husband and I automatically agreed that the gene therapy sounded like the most promising option for her to have a life.”
‘An hour that changed our whole life’
Because the companies developing Zolgensma had already applied for FDA approval, Leigh would need to request special expanded access from the FDA to use the therapy in Arabella. She prepared Sarah and Vitaliy for the lengthy process ahead. Both were surprised when all the approvals came through within a month.
“We were still working through what the diagnosis meant for our family, so when we got the news that Arabella was approved for the gene therapy, we were in absolute shock,” Sarah said. “We were thankful Dr. Leigh made it happen so fast.”
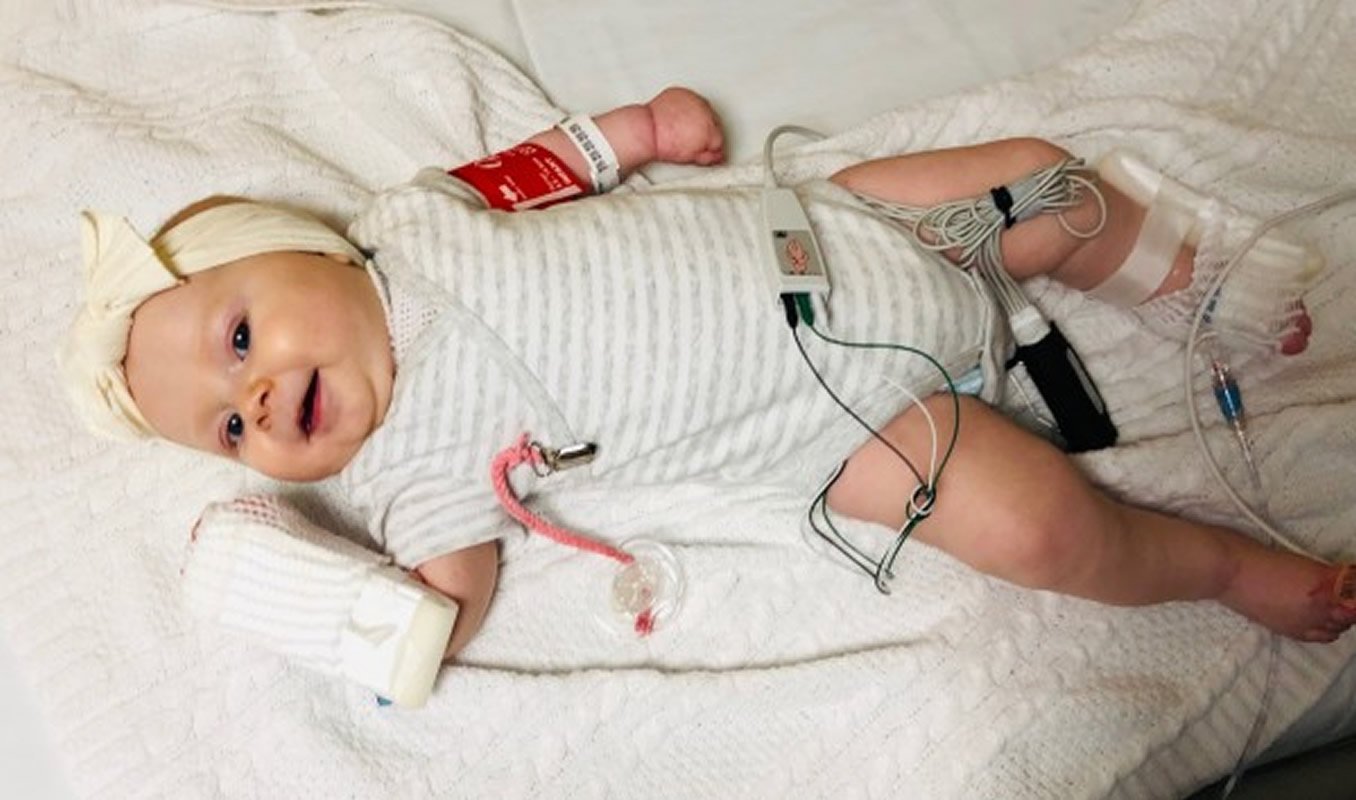
The family made plans for Arabella to receive her dose of Zolgensma at Seattle Children’s in February 2019. In a seemingly unremarkable infusion, the therapy transferred a functional copy of the SMN1 gene to her affected motor neurons by way of a virus shell engineered specifically for the purpose of delivery.
“While it was just a simple IV, that one hour changed our whole life,” Sarah said.
Once the SMN1 gene reached Arabella’s cells, it supplemented those cells’ own production of SMN protein to improve motor neuron function.
“That’s the beauty of Zolgensma,” Leigh said. “It’s one dose and then it self-replicates.”
Leigh believes the one-time treatment of Zolgensma offers is a benefit over Spinraza, which has produced similar outcomes, but requires regular doses administered through lumbar puncture over the patient’s lifetime.
A miracle everyday
Three months after receiving Zolgensma, Arabella’s parents say it’s hard to put into words the feeling they have as they watch Arabella hit milestones they never dreamed would be possible.
“It’s like looking at a miracle happening before your eyes every single day,” Vitaliy said. “She’s so full of life.”
Since receiving the gene therapy, Arabella, now 7 months old, has accomplished skills once unheard of for babies with SMA. Her parents remember the first time they noticed her holding her head up on her own. She was in her infant swing, and instead of her head flopping to one side like it had always done, this day, she was holding it straight against the movement of the swing.
“We just watched her do that over and over again,” Sarah said. “We were in awe to see her little body working as she was trying to lift her head.”
Leigh noticed a similar newfound strength in Arabella’s most recent follow up appointment. She did a test of leg strength by holding Arabella upright to see if she would push off the exam table underneath her.
“You could feel her grip the table for one, maybe two, seconds,” she said. “Afterward, her mom repeated the same test at home and said she thought she felt Arabella standing like this for longer than five seconds.”
Arabella is eating stage 2 baby food which means she doesn’t have any problem swallowing and she will not need a feeding tube. Since all infants with SMA type 1 require feeding tubes, this is a huge step forward for Arabella.
‘She’s blossoming now’
Leigh says this kind of improvement is transforming the way she and other specialists in the field manage care for babies with SMA type 1.
“Before we could only really offer palliative care,” she said. “Now, the hope is that they get enough strength and skeletal tone that they can bear weight and one day walk.”
In addition to follow up appointments with Leigh, Arabella goes to weekly therapy sessions at Seattle Children’s North Clinic in Everett near her home in Lynnwood, Washington to build her strength and mobility.
“We’ve noticed a lot of improvement since she’s started the therapy,” Sarah said. “At first you think, ‘Oh, it’s just physical therapy,’ but it’s almost like she’s blossoming now.”
Giving families the choice of Zolgensma
Even with the FDA approval, there remain unanswered questions about Zolgensma, such as how much of its cost insurers are willing to cover and whether the drug manufacturing process can keep up with the demand for the drug. Leigh says she and many others have been working behind the scenes to address these questions because she hopes every family will have the opportunity to consider the gene therapy as an option.
“At the end of the day, I think all the individuals involved with making this drug possible, myself especially, just want to do what’s best for the child,” she said.
She sees Seattle Children’s contributing to ongoing research efforts to improve the delivery of Zolgensma and evaluate its use in older patients with SMA type 2 and 3.
She also wants to ensure that every baby is screened as early as possible for SMA and is advocating for the addition of SMA to Washington state’s newborn screening panel. The Washington State Board of Health will review the recommendation unanimously approved by the committee Leigh participated in this spring at their next meeting in June.
“Now that we have options to treat SMA, early detection can make the future possible for these babies,” Leigh said. “It just makes sense that every child born should have the chance to receive screening for SMA.”
For families of children like Arabella, it is all about the future.
“Gene therapy sounded like something from a science fiction movie and I’m not sure we’ll ever completely understand how it works,” Vitaliy said. “But the future is now, so we decided to trust the process and let the doctors do what they do best to let Arabella have the best life possible.”
Source:
Seattle Children’s Hospital
Media Contacts:
Lindsay Kurs – Seattle Children’s Hospital
Image Source:
The image is credited to Seattle Children’s Hospital.