Summary: Researchers have identified a potential molecular mechanism that causes Fragile X syndrome in the developing fetal brain.
Source: Tohoku University
Researchers at Tohoku University have revealed further insight into the fetal development of our brain and the potential causes of Fragile X syndrome (FSX).
During brain development, the fetal period is vital in creating neurons from neural stem cells to build up a functional adult brain. Any impairment in the developmental program could result in severe defects in the brain.
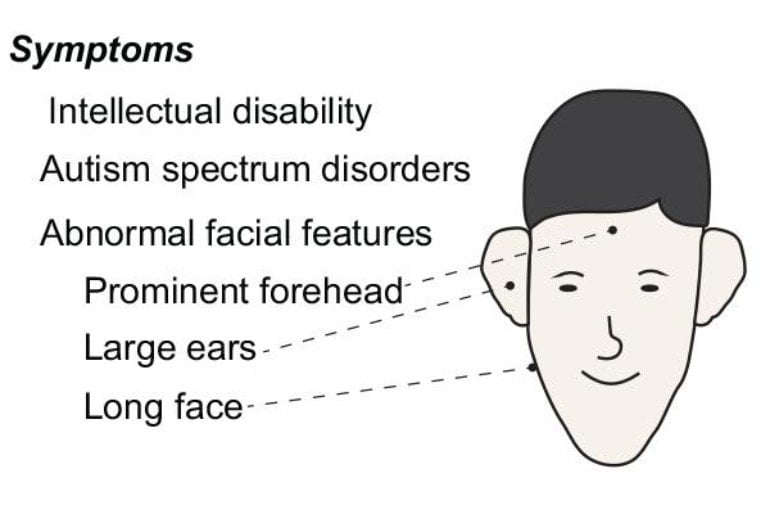
FSX is a genetic disorder characterized by intellectual disability and autistic symptoms. Children with FSX will generally suffer from developmental delays as well as social and behavioral problems.
FSX patients have a defect in the fragile X mental retardation 1 (FMR1) gene, a gene that codes for the fragile X mental retardation protein (FNRP)–the critical factor in normal brain development.
“Our study illustrated the possible molecular mechanism that causes FXS in the fetal brain and furthers our understanding of hereditary developmental disorders in the brain’s developmental stage,” said Noriko Osumi, professor at the Department of Developmental Neuroscience, Tohoku University Graduate School of Medicine.
Using next-generation sequencing, Osumi and her team identified hundreds of FMRP regulated molecules in mice fetal brains. These molecules were associated not only with neurogenesis but also autism and intellectual disability.
Their findings showed that specific groups of molecules were involved in the intracellular signaling pathways such as Ras/MAPK, Wnt/β-catenin, and mTOR.
The mTOR activity was significant in the fetal brain of FMR1 deficient mice, suggesting that increased mTOR activity may lead to abnormal proliferation and differentiation of neural stem cells in the fetal brain. Ultimately, these molecular mechanisms could be responsible for developing the brain during the fetal period and contribute to the causes of FXS.
The research team hopes this new information will serve as an essential resource for future studies of neurodevelopmental disorders.
About this Fragile X research news
Source: Tohoku University
Contact: Noriko Osumi – Tohoku University
Image: The image is credited to Tohoku University Graduate School of Medicine
Original Research: Closed access.
“Identification of FMRP target mRNAs in the developmental brain: FMRP might coordinate Ras/MAPK, Wnt/β-catenin, and mTOR signaling during corticogenesis” by Cristine R. Casingal, Takako Kikkawa, Hitoshi Inada, Yukio Sasaki & Noriko Osumi. Molecular Brain
Abstract
Identification of FMRP target mRNAs in the developmental brain: FMRP might coordinate Ras/MAPK, Wnt/β-catenin, and mTOR signaling during corticogenesis
Corticogenesis is one of the most critical and complicated processes during embryonic brain development. Any slight impairment in corticogenesis could cause neurodevelopmental disorders such as Fragile X syndrome (FXS), of which symptoms contain intellectual disability (ID) and autism spectrum disorder (ASD). Fragile X mental retardation protein (FMRP), an RNA-binding protein responsible for FXS, shows strong expression in neural stem/precursor cells (NPCs) during corticogenesis, although its function during brain development remains largely unknown. In this study, we attempted to identify the FMRP target mRNAs in the cortical primordium using RNA immunoprecipitation sequencing analysis in the mouse embryonic brain. We identified 865 candidate genes as targets of FMRP involving 126 and 118 genes overlapped with ID and ASD-associated genes, respectively. These overlapped genes were enriched with those related to chromatin/chromosome organization and histone modifications, suggesting the involvement of FMRP in epigenetic regulation. We further identified a common set of 17 FMRP “core” target genes involved in neurogenesis/FXS/ID/ASD, containing factors associated with Ras/mitogen-activated protein kinase, Wnt/β-catenin, and mammalian target of rapamycin (mTOR) pathways. We indeed showed overactivation of mTOR signaling via an increase in mTOR phosphorylation in the Fmr1 knockout (Fmr1 KO) neocortex. Our results provide further insight into the critical roles of FMRP in the developing brain, where dysfunction of FMRP may influence the regulation of its mRNA targets affecting signaling pathways and epigenetic modifications.