Researchers have identified a new set of genes that may be responsible for the two most common and disabling neurological conditions, stroke and dementia.
The study, which appears in the journal Lancet Neurology, may help researchers better understand, treat and prevent ischemic and hemorrhagic stroke, and perhaps Alzheimer’s disease and other dementias.
Stroke is the leading neurological cause of death and disability worldwide. Previous studies have looked mainly at genes causing atherosclerosis and genes affecting the function of platelets and clotting processes as risk factors for ischemic stroke (clot obstructing blood flow to the brain). A different set of genes have been associated with hemorrhagic stroke (bleeding into the brain).
Researchers from Boston University School of Medicine looked for new stroke genes using genome wide association as well as meta-analysis. They identified a new gene called FOXF2 which increased the risk of having a stroke due to small vessel disease in the brain. No previous study has identified a gene for the common type of small vessel disease stroke although some genes associated with familial small vessel diseases such as CADASIL are known.
“Our research has identified a gene affecting another type of ischemic stroke, due to small vessel disease, and also suggests some genes may be associated with both ischemic and hemorrhagic stroke and may act through a novel pathway affecting pericytes, a type of cell in the wall of small arteries and capillaries. Unraveling the mechanisms of small vessel disease is essential for the development of therapeutic and preventive strategies for this major cause of stroke,” explained corresponding author Sudha Seshadri, MD, professor of neurology at BUSM.
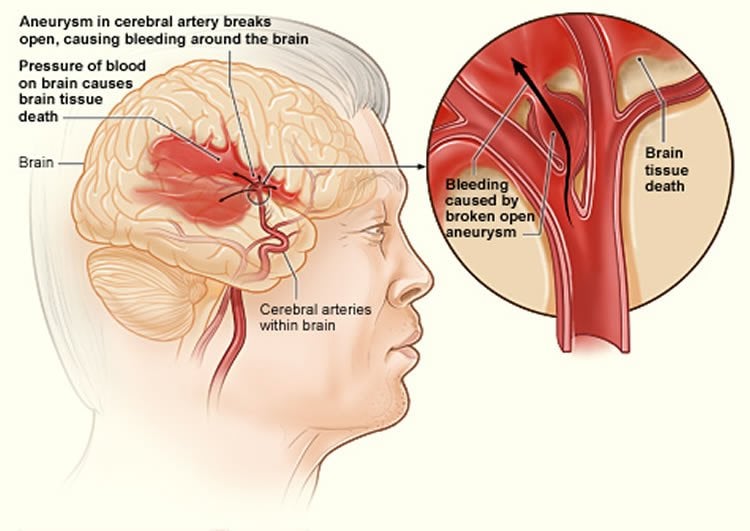
According to the researchers small vessel disease not only causes stroke but is also a major contributor to dementia risk, and is associated with gait problems and depression. “Hence, it is exciting that we are beginning to better understand the cause of this very important and poorly understood type of stroke,” she added.
The study was carried out in collaboration between researchers at Boston University School of Medicine, Bordeaux University, Washington University, the University of Calgary, Gothenburg University and many others.
Funding: Funding for this study was provided by the National Institute of Neurological Disorders and Stroke: NS017950, the National Institute on Aging: AG033193 and the National Heart, Lung and Blood Institute.
Source: Gina DiGravio – Boston University Medical Center
Image Credit: The image is credited to the NHI and is in the public domain.
Original Research: Abstract for “Identification of additional risk loci for stroke and small vessel disease: a meta-analysis of genome-wide association studies” by Neurology Working Group of the Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium Stroke Genetics Network (SiGN) International Stroke Genetics Consortium (ISGC) in Lancet Neurology. Published online April 7 2016 doi:10.1016/S1474-4422(16)00102-2
Abstract
Identification of additional risk loci for stroke and small vessel disease: a meta-analysis of genome-wide association studies
Background
Genetic determinants of stroke, the leading neurological cause of death and disability, are poorly understood and have seldom been explored in the general population. Our aim was to identify additional loci for stroke by doing a meta-analysis of genome-wide association studies.
Methods
For the discovery sample, we did a genome-wide analysis of common genetic variants associated with incident stroke risk in 18 population-based cohorts comprising 84 961 participants, of whom 4348 had stroke. Stroke diagnosis was ascertained and validated by the study investigators. Mean age at stroke ranged from 45·8 years to 76·4 years, and data collection in the studies took place between 1948 and 2013. We did validation analyses for variants yielding a significant association (at p<5 × 10−6) with all-stroke, ischaemic stroke, cardioembolic ischaemic stroke, or non-cardioembolic ischaemic stroke in the largest available cross-sectional studies (70 804 participants, of whom 19 816 had stroke). Summary-level results of discovery and follow-up stages were combined using inverse-variance weighted fixed-effects meta-analysis, and in-silico lookups were done in stroke subtypes. For genome-wide significant findings (at p<5 × 10−8), we explored associations with additional cerebrovascular phenotypes and did functional experiments using conditional (inducible) deletion of the probable causal gene in mice. We also studied the expression of orthologs of this probable causal gene and its effects on cerebral vasculature in zebrafish mutants.
Findings
We replicated seven of eight known loci associated with risk for ischaemic stroke, and identified a novel locus at chromosome 6p25 (rs12204590, near FOXF2) associated with risk of all-stroke (odds ratio [OR] 1·08, 95% CI 1·05–1·12, p=1·48 × 10−8; minor allele frequency 21%). The rs12204590 stroke risk allele was also associated with increased MRI-defined burden of white matter hyperintensity—a marker of cerebral small vessel disease—in stroke-free adults (n=21 079; p=0·0025). Consistently, young patients (aged 2–32 years) with segmental deletions of FOXF2 showed an extensive burden of white matter hyperintensity. Deletion of Foxf2 in adult mice resulted in cerebral infarction, reactive gliosis, and microhaemorrhage. The orthologs of FOXF2 in zebrafish (foxf2b and foxf2a) are expressed in brain pericytes and mutant foxf2b−/− cerebral vessels show decreased smooth muscle cell and pericyte coverage.
Interpretation
We identified common variants near FOXF2 that are associated with increased stroke susceptibility. Epidemiological and experimental data suggest that FOXF2 mediates this association, potentially via differentiation defects of cerebral vascular mural cells. Further expression studies in appropriate human tissues, and further functional experiments with long follow-up periods are needed to fully understand the underlying mechanisms.
Funding
NIH, NINDS, NHMRC, CIHR, European national research institutions, Fondation Leducq.
“Identification of additional risk loci for stroke and small vessel disease: a meta-analysis of genome-wide association studies” by Neurology Working Group of the Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium Stroke Genetics Network (SiGN) International Stroke Genetics Consortium (ISGC) in Lancet Neurology. Published online April 7 2016 doi:10.1016/S1474-4422(16)00102-2







