Summary: Selective estrogen treatments appear to reduce symptoms of Parkinson’s disease in male mice. The study sheds light on sex-based differences associated with the disease and may provide new therapeutic avenues for treatment.
Source: SfN
Brain-selective estrogen treatment improves the symptoms of Parkinson’s disease in male mice, according to new research published in JNeurosci. These findings may help explain the sex differences in Parkinson’s disease and could lead to estrogen-based treatments.
Parkinson’s disease is characterized by the death of neurons involved in movement, which may be partially caused by gene mutations for the protein α-synuclein. The mutated, shorter form of the protein clusters in neurons, resulting in their death, while the longer form resists clumping.
Estrogen is thought to protect movement neurons from Parkinson’s disease, but how is unknown. Since the patients more susceptible to Parkinson’s disease — men and post-menopausal women — have low estrogen levels, estrogen treatment might be an effective way to delay and reduce symptoms.
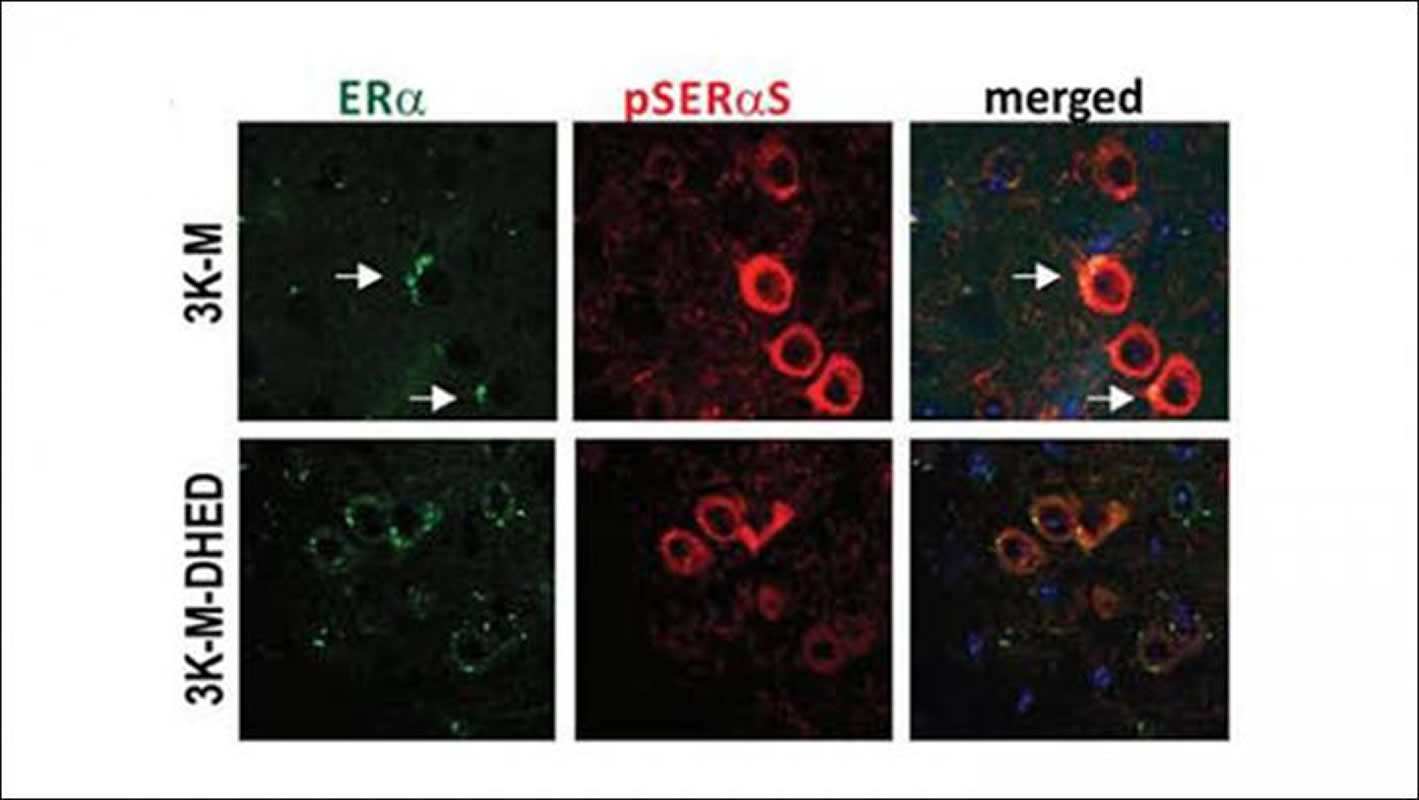
Silke Nuber and colleagues at Harvard Medical School treated mouse models of Parkinson’s disease with brain-selective estrogen and compared the motor performance of males and females before and after treatment. The female mice showed less severe symptoms at a later age, but estrogen still improved their symptoms. In male mice, the estrogen treatment reduced α-synuclein breakdown and buildup and helped with severe symptoms, suggesting that estrogen could be a viable treatment option for Parkinson’s patients with low estrogen levels.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Rajsombath et al., JNeurosci 2019.
Original Research: Closed access
“Female Sex and Brain-Selective Estrogen Benefit α-Synuclein Tetramerization and the PD-Like Motor Syndrome in 3K Transgenic Mice”. Molly M Rajsombath, Alice Y Nam, Maria Ericsson and Silke Nuber.
Journal of Neuroscience. doi:10.1523/JNEUROSCI.0313-19.20194
Abstract
Female Sex and Brain-Selective Estrogen Benefit α-Synuclein Tetramerization and the PD-Like Motor Syndrome in 3K Transgenic Mice
Many studies report a higher risk for Parkinson’s disease (PD) and younger age of onset in men. This, and the fact that the neuropathological process underlying PD-symptoms may begin before menopause, suggest that estrogen-based hormone therapy could modify this higher risk in males. However, the effects of female sex or estrogen on α-synuclein (αS) homeostasis and related PD neuropathology remain unknown. Here, we used an αS tetramer-abrogating mouse model of PD (3K) that amplifies the familial E46K PD mutation to investigate the effects of female sex and brain-selective estrogen treatment on αS tetramerization and solubility, formation of vesicle-rich αS+ aggregates, dopaminergic and cortical fiber integrity, and associated motor deficits. In male 3K mice, the motor phenotype became apparent at ∼10 wk and increased to age 6 mo, paralleled by PD-like neuropathology, whereas 3K females showed a significant delay in onset. At 6 mo, this beneficial phenotypic effect in 3K females was associated with a higher αS tetramer-to-monomer (T:M) ratio and less decrease in dopaminergic and cortical fiber length and quantity. Brain-selective estrogen treatment in symptomatic 3K males significantly increased the T:M ratio, turnover by autophagy of aggregate-prone monomers, and neurite complexity of surviving DAergic and cortical neurons, in parallel with benefits in motor performance. Our findings support an upstream role for αS tetramer loss in PD phenotypes and a role for estrogen in mitigating PD-like neuropathology in vivo. Brain-selective estrogen therapy may be useful in delaying or reducing PD symptoms in men and postmenopausal women.
SIGNIFICANCE STATEMENT
The mechanisms responsible for the male-to-female preponderance in PD are not well understood, yet important for treatment efficacy. We previously showed that abrogating native αS tetramers produces a close PD model, including dopaminergic and cortical fiber loss and a progressive motor disorder responsive to L-DOPA. Here, we analyzed sex and use DHED treatment of symptomatic 3K males and demonstrate the beneficial effects of female sex on PD-like neuropathology can be reinstated by elevating estrogen in the male brain. The study provides evidence that 17β-estradiol restores the tetramer-to-monomer rate by autophagy turnover of excess αS monomers, vesicle and fiber integrity in brain regions critically involved in motor behavior. These data provide the basis for understanding sex differences in αS homeostasis and the development of therapeutic approaches to treating men and postmenopausal women with PD.






