Summary: Environmental enrichment can help correct mismapped neurons in the visual pathways of mice.
Source: SfN
Environmental enrichment can partially correct miswired neurons in the visual pathway, according to research in mice recently published in eNeuro.
During normal development, neurons exit the retina and form connections in specific area of the lateral geniculate nucleus, a part of the brain involved in visual processing. In mice with a genetic mutation, the neurons land throughout the entire lateral geniculate nucleus in a thin, dispersed line. Such mapping errors lead to vision issues in adulthood.
Eggins et al. raised mice with genetic mapping errors in a standard cage or in an enriched environment with a variety of toys and stimulation. The mice living in an enriched environment from birth displayed less mapping errors in the lateral geniculate nucleus than those raised in standard cages. Mice without enrichment until adolescence or adulthood had the same mapping errors as the mice that lived in a standard cage.
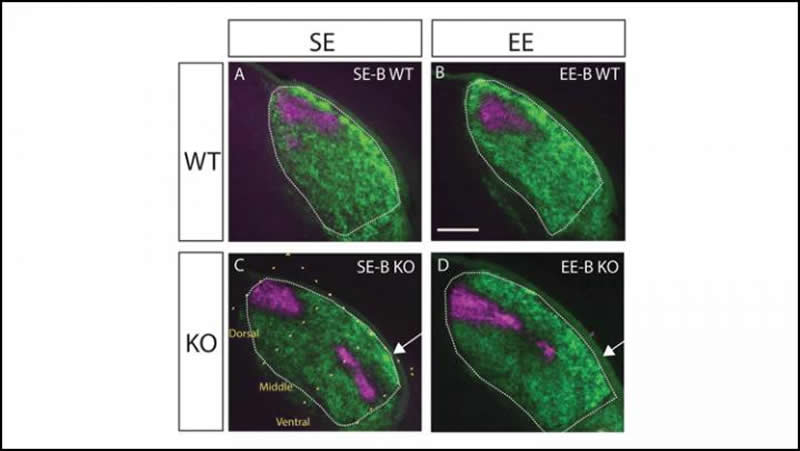
Enrichment can counteract mapping errors, but within a strict critical period after birth. These findings point to the potential of using early childhood enrichment as a treatment for neurodevelopmental disorders rooted in miswired neurons, such as autism.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Eggins et al., eNeuro 2019.
Original Research: Open access
“Environmental Enrichment Partially Repairs Subcortical Mapping Errors in Ten-m3 Knockout Mice During an Early Critical Period”. Peta Eggins, James Blok, Justin Petersen, Larissa Savvas, Lara Rogerson-Wood, Hannan Mansuri, Atomu Sawatari and Catherine A. Leamey.
eNeuro doi:10.1523/ENEURO.0478-18.2019.
Abstract
Environmental Enrichment Partially Repairs Subcortical Mapping Errors in Ten-m3 Knockout Mice During an Early Critical Period
Environmental enrichment (EE) has been shown to improve neural function via the regulation of cortical plasticity. Its capacity to induce functional and/or anatomical repair of miswired circuits is unknown. Ten-m3 knockout (KO) mice exhibit a highly-stereotyped and profound miswiring of ipsilateral retinogeniculate axons and associated deficits in binocularly-mediated visual behaviour. We determined whether, and when, EE can drive the repair of subcortical wiring deficits by analysing Ten-m3 KO and wildtype mice that were enriched for 6 weeks from adulthood, weaning or birth in comparison to standard-housed controls. Six weeks of EE initiated from birth, but not later, induced a significant reduction in the area occupied by ipsilateral retinogeniculate terminals in KOs enriched from birth. No EE-induced correction of mistargeted axons was observed at postnatal day 7, indicating that this intervention impacts pruning rather than initial targeting of axons. This reduction was most prominent in the ventrolateral region of the dorsal lateral geniculate nucleus, suggesting a preferential pruning of the most profoundly mistargeted axons. EE can thus partially repair a specific, subcortical axonal wiring deficit, but only during an early, developmentally-restricted time-window.
Significance Statement
The incorrect wiring of neural circuits can lead to profound disability. Using a mouse model which exhibits a marked miswiring of neural projections to subcortical relay centres of the brain, we show that positive modulation (enrichment) of the environment during the first few postnatal weeks can induce the pruning of aberrant neural projections. The capacity for environmental enrichment to correct miswired projections diminished prior to adolescence, suggesting that the enrichment can only drive these changes during an early critical period. This expands our knowledge of the capacity for enrichment to induce repair of neural circuits. The demonstration of dramatic effects on targeting of neural projections over a restricted period may have implications for the development of therapies for neurodevelopmental disorders.






