Researchers have thus far been unable to define how dopamine influences cocaine reward. A recent report published in Neuron has shown that cocaine administration increases dopamine levels in the striatum, activating a signaling pathway that was previously unknown.
Dopamine activates a protein called PKA in striatal neurons, which activates many additional substrates to regulate neuronal excitability and control behavior. However, the identities of these PKA substrates are not known. Recently, a research team at Nagoya University has uncovered the answers.
“We stimulated PKA in mouse brain slices to activate these unknown substrates,” explains corresponding author Kozo Kaibuchi of the University of Nagoya’s Department of Cell Pharmacology. His research team was able to extract these activated proteins from brain slices and identify them. “Using this screening approach, we identified more than 100 candidate substrates of PKA,” continues Kaibuchi.
One of these novel candidates was Rasgrp2, a protein that is highly expressed in striatal neurons. Rasgrp2 positively regulates another protein called Rap1 and the authors were intrigued to find out whether Rap1 was activated by Rasgrp2 in striatal neurons. Through a range of experiments, the Nagoya research group found the answer; they demonstrated that cocaine treatment increased the phosphorylation of Rasgrp2 by PKA, which in turn activated Rap1 in striatal neurons.
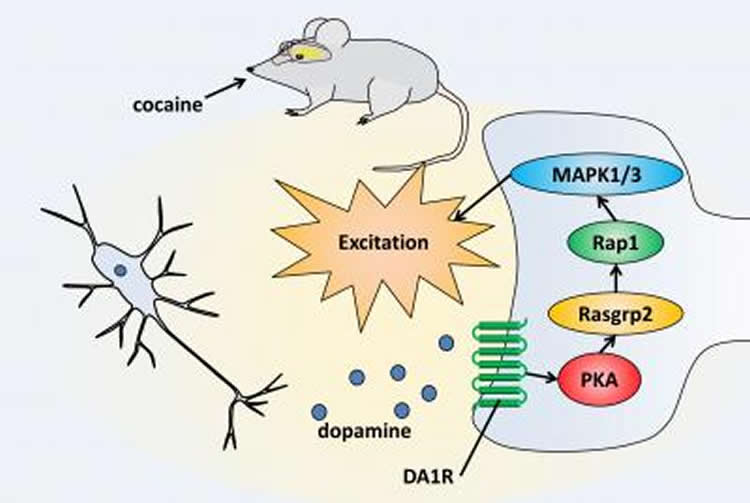
“We speculated that the Rap1 pathway is involved in the regulation of neuronal functions by dopamine,” says lead author Taku Nagai at the university’s Department of Neuropsychcopharmacology. By measuring neuronal activity directly in mouse brain slices, Nagai and colleagues discovered that activated Rap1 can increase the activity of dopaminergic neurons in the striatum. In addition, the rewarding effects of cocaine were higher when Rap1 was activated in mouse brains.
“When dopamine levels increase due to cocaine administration, striatal neurons become more excitable, resulting in enhanced responses to excitatory input from other brain regions,” explains Nagai. “This increases the sensitivity of mice to cocaine reward.”
Many of the 100 candidate PKA substrates identified were previously unknown as components of dopamine signaling. Therefore in the future, these findings will significantly contribute to our understanding of the different roles of dopamine in many brain functions.
Funding: Funding was provided by Japan Agency for Medical Research and Development.
Source: Koomi Sung – Nagoya University
Image Credit: Image is credited to Nagoya University.
Original Research: Abstract for “Phosphoproteomics of the Dopamine Pathway Enables Discovery of Rap1 Activation as a Reward Signal In Vivo” by Taku Nagai, Shinichi Nakamuta, Keisuke Kuroda, Sakura Nakauchi, Tomoki Nishioka, Tetsuya Takano, Xinjian Zhang, Daisuke Tsuboi, Yasuhiro Funahashi, Takashi Nakano, Junichiro Yoshimoto, Kenta Kobayashi, Motokazu Uchigashima, Masahiko Watanabe, Masami Miura, Akinori Nishi, Kazuto Kobayashi, Kiyofumi Yamada, Mutsuki Amano, and Kozo Kaibuchi in Neuron. Published online January 21 2016 doi:10.1016/j.neoron2015.12.019
Abstract
Phosphoproteomics of the Dopamine Pathway Enables Discovery of Rap1 Activation as a Reward Signal In Vivo
Highlights
•Phosphoproteomic analysis identified PKA substrates downstream of D1R
•More than 100 candidate substrates of PKA were found, including Rap1 GEF (Rasgrp2)
•PKA-mediated Rasgrp2 phosphorylation enhances its GEF activity on Rap1
•Rap1 activation regulates neuronal excitability and cocaine reward-related behavior
Summary
Dopamine (DA) type 1 receptor (D1R) signaling in the striatum presumably regulates neuronal excitability and reward-related behaviors through PKA. However, whether and how D1Rs and PKA regulate neuronal excitability and behavior remain largely unknown. Here, we developed a phosphoproteomic analysis method to identify known and novel PKA substrates downstream of the D1R and obtained more than 100 candidate substrates, including Rap1 GEF (Rasgrp2). We found that PKA phosphorylation of Rasgrp2 activated its guanine nucleotide-exchange activity on Rap1. Cocaine exposure activated Rap1 in the nucleus accumbens in mice. The expression of constitutively active PKA or Rap1 in accumbal D1R-expressing medium spiny neurons (D1R-MSNs) enhanced neuronal firing rates and behavioral responses to cocaine exposure through MAPK. Knockout of Rap1 in the accumbal D1R-MSNs was sufficient to decrease these phenotypes. These findings demonstrate a novel DA-PKA-Rap1-MAPK intracellular signaling mechanism in D1R-MSNs that increases neuronal excitability to enhance reward-related behaviors.
“Phosphoproteomics of the Dopamine Pathway Enables Discovery of Rap1 Activation as a Reward Signal In Vivo” by Taku Nagai, Shinichi Nakamuta, Keisuke Kuroda, Sakura Nakauchi, Tomoki Nishioka, Tetsuya Takano, Xinjian Zhang, Daisuke Tsuboi, Yasuhiro Funahashi, Takashi Nakano, Junichiro Yoshimoto, Kenta Kobayashi, Motokazu Uchigashima, Masahiko Watanabe, Masami Miura, Akinori Nishi, Kazuto Kobayashi, Kiyofumi Yamada, Mutsuki Amano, and Kozo Kaibuchi in Neuron. Published online January 21 2016 doi:10.1016/j.neoron2015.12.019






