Summary: Researchers successfully turned skin cells from Parkinson’s patients into dopaminergic neurons by introducing a combination of neural-inducing genes into the skin cells.
Source: International Society for Stem Cell Research
The possibility to make virtually all cell types of the human body from induced pluripotent stem cells (iPSCs), which are embryonic-like cells generated from a patient’s skin, in a process called reprogramming, has opened new avenues for disease modeling in the lab.
However, one shortcoming of this technique is that the donor age-specific cellular features are erased during reprogramming, so that cells made from iPSCs typically resemble cells in the human embryo or fetus rather than cells in the adult or aged human individual.
Neurodegenerative diseases like Parkinson’s disease (PD) mainly affect older individuals and are therefore difficult to model with PSC-derived neurons, which lack many defining features of aged neurons.
According to a recent report from Janelle Drouin-Ouellet (University of Montreal, Canada), Malin Parmar (University of Lund, Sweden) and colleagues in the journal Stem Cell Reports, one way of preserving the aged characteristics of neurons is to make them directly from the patient’s skin, without an iPSC intermediate.
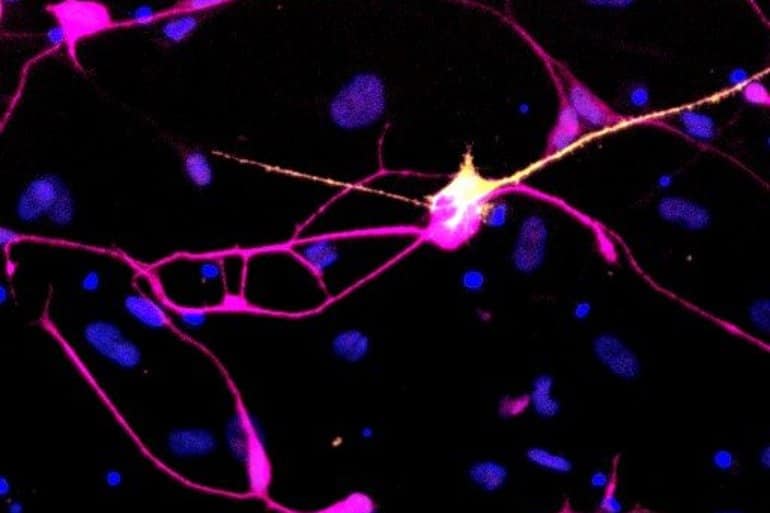
The researchers succeeded in turning skin cells from PD patients into so-called dopaminergic (DA) neurons, which are the type of neurons progressively lost in PD, by introducing a specific combination of neural-inducing genes into the skin cells.
In contrast to the PD cells generated from iPS cells, this process of generating DA neurons directly from the skin cells preserved the aged genetic, epigenetic, and metabolic characteristics of the donor age.
When compared to aged DA neurons from healthy skin donors, neurons from PD patients featured PD-specific cellular defects, which now could be modeled for the first time from sporadic PD patients which lack a known genetic mutation.
With this new tool, the researchers hope to model PD-related neuronal defects from a much larger cohort of PD patients with the aim to identify identity causes of the disease as well as potential future therapies.
About this genetics and Parkinson’s disease research news
Author: Press Office
Source: International Society for Stem Cell Research
Contact: Press Office – International Society for Stem Cell Research
Image: The image is credited to Janelle Drouin-Ouellet, University of Montreal
Original Research: Open access.
“Age-related pathological impairments in directly reprogrammed dopaminergic neurons derived from patients with idiopathic Parkinson’s disease” by Janelle Drouin-Ouellet et al. Stem Cell Reports
Abstract
Age-related pathological impairments in directly reprogrammed dopaminergic neurons derived from patients with idiopathic Parkinson’s disease
Highlights
- Cell reprogramming model by which to study age neuronal features of idiopathic PD
- Reprogrammed neurons exhibit disease-relevant impairments in autophagy
- Some phenotypes were specific to the dopaminergic neurons
- iDANs show age-related autophagy impairments leading to alpha-synuclein accumulation
Summary
We have developed an efficient approach to generate functional induced dopaminergic (DA) neurons from adult human dermal fibroblasts. When performing DA neuronal conversion of patient fibroblasts with idiopathic Parkinson’s disease (PD), we could specifically detect disease-relevant pathology in these cells.
We show that the patient-derived neurons maintain age-related properties of the donor and exhibit lower basal chaperone-mediated autophagy compared with healthy donors. Furthermore, stress-induced autophagy resulted in an age-dependent accumulation of macroautophagic structures.
Finally, we show that these impairments in patient-derived DA neurons leads to an accumulation of phosphorylated alpha-synuclein, the classical hallmark of PD pathology. This pathological phenotype is absent in neurons generated from induced pluripotent stem cells from the same patients.
Taken together, our results show that direct neural reprogramming can be used for obtaining patient-derived DA neurons, which uniquely function as a cellular model to study age-related pathology relevant to idiopathic PD.






