Summary: Researchers have developed a compound that blocks the FKBP51 protein, which helps reduce chronic pain, provides positive outcomes for diet-induced obesity and improves symptoms of depression.
Source: American Chemical Society
Major depression, obesity, and chronic pain are all linked to the effects of one protein, called “FK506-binding protein 51,” or FKBP51. Until now, efforts to inhibit this target have been hampered by the difficulty of finding something specific enough to do the job and not affect similar proteins. Now a research group has developed a highly selective compound that can effectively block FKBP51 in mice, relieving chronic pain and having positive effects on diet-induced obesity and mood. The new compound also could have applications in alcoholism and brain cancer.
The researchers will present their results today at the American Chemical Society (ACS) Spring 2019 National Meeting & Exposition. ACS, the world’s largest scientific society, is holding the meeting here through Thursday. It features nearly 13,000 presentations on a wide range of science topics.
“The FKBP51 protein plays an important role in depression, obesity, diabetes, and chronic pain states,” says Felix Hausch, Ph.D., the project’s principal investigator. “We developed the first highly potent, highly selective FKBP51 inhibitor, called SAFit2, which is now being tested in mice. Inhibition of FKBP51 could thus be a new therapeutic option to treat all of these conditions.”
Hausch, who is at the Technical University of Darmstadt, started the project when studies were published linking the protein to depression.
“I was intrigued by the peculiar regulatory role it seemed to play in cells,” he says. “And there was a known natural product that could serve as a starting point. Collectively, this looked like an interesting protein to work on.”
FKBP51 is expressed in multiple places throughout the body, such as the brain, skeletal muscle tissue and fat. It also has multiple effects. For example, the protein can restrict the uptake of glucose and the browning of fat, so that the body stores fat instead of burning it. It also affects stress responses. So, Hausch and his colleagues figured that blocking this protein could be the key to developing drugs to treat a variety of conditions.
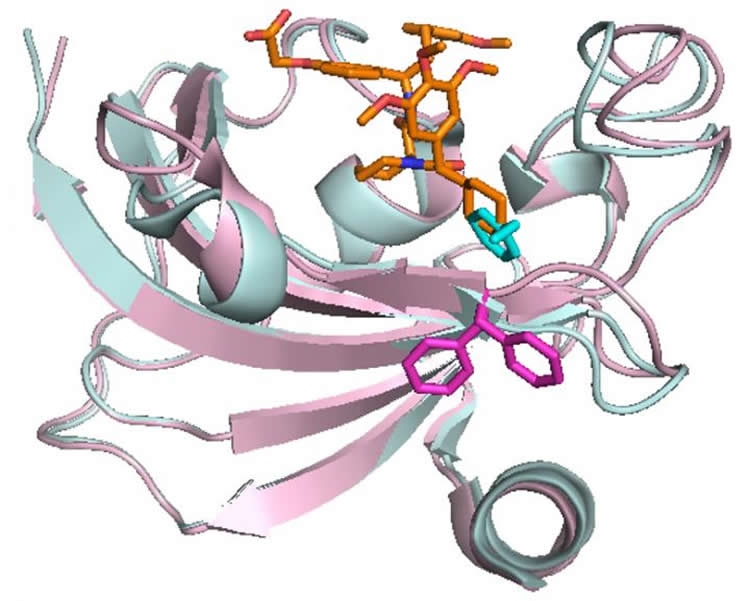
But FKBP51 looks a lot like its closest protein cousin, FKBP52. “These two proteins are very similar in structure, but they are doing opposing things in cells,” Hausch says. “We have this yin-yang situation. Selectivity between these two proteins is thought to be crucial, but this is hard to achieve since the two proteins are so similar. We discovered that FKBP51 can change its shape in a way that FKBP52 can’t, and this allowed the development of highly selective inhibitors.”
The researchers have now used nuclear magnetic resonance techniques to detect a previously hidden binding site in FKBP51. The approach could help other researchers identify similar “cryptic” binding sites in challenging drug targets in the future, Hausch says.
His team is now testing SAFit2, the lead FKBP51 inhibitor they developed from these studies, in animals. “It indeed helps mice cope better in stressful situations,” Hausch says. In mice, SAFit2 reduced stress hormone levels, promoted more active stress coping, was synergistic with antidepressants, protected against weight gain, helped normalize glucose levels and reduced pain in three animal models.
According to Hausch, much more needs to be done to get FKBP1 inhibitors to the point where they could be used as a drug molecule in human testing. In the meantime, the team is also exploring FKBP51 inhibitors in other applications. So far, the group has conducted a number of mouse studies on the involvement of FKBP51 in alcoholism, but results are still preliminary. In addition, Hausch points out that certain types of glioblastoma tumors overexpress FKBP51. He hopes that this result indicates FKBP51 inhibitors could be used in cancer treatment when patients’ tumors mutate beyond current drugs’ capacity to treat them. “We may be able to resensitize them to different types of chemotherapy using these specific inhibitors,” he says.
Funding: The researchers acknowledge support and funding from European Research Area Industrial Biotechnology, the Federal Ministry of Education and Research and the Pioneer Fund of the Technical University of Darmstadt.
Source:
American Chemical Society
Media Contacts:
Katie Cottingham, Ph.D. – American Chemical Society
Image Source:
The image is credited to Felix Hausch.
Original Research: The findings will be presented at the American Chemical Society 257th National Meeting in Orlando, Florida, USA.







