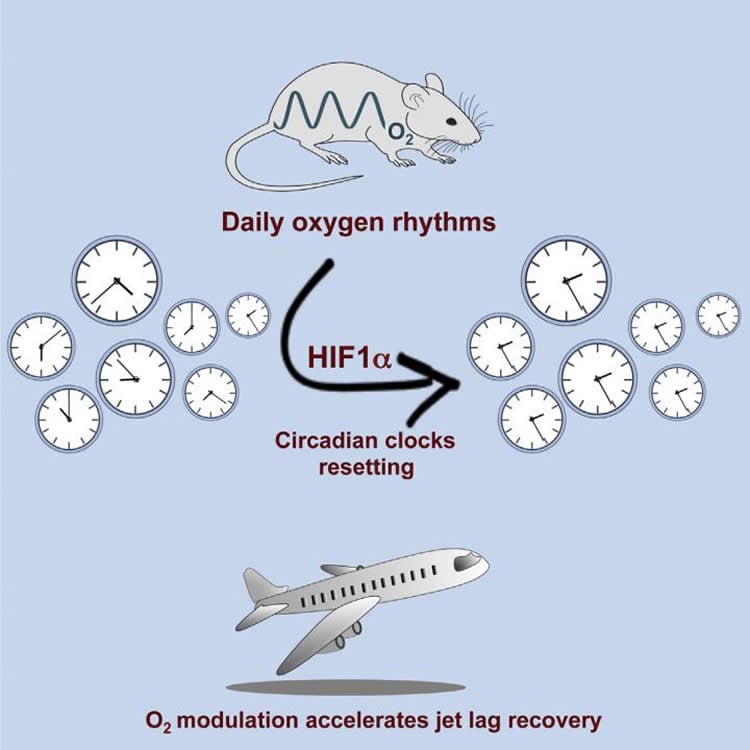
Summary: Researchers report variations in surrounding oxygen levels can reset the circadian clock in mice.
Source: Cell Press.
We might not think of our circadian clock until we are jetlagged, but scientists continue to puzzle over what drives our biological timepiece. Now, a study published October 20 in Cell Metabolism has found that variations in surrounding oxygen levels can reset circadian clocks of mice. If confirmed in humans, the research could help inform how airlines moderate cabin air pressure.
Presently, light, food, and temperature are the best known cues that can influence circadian rhythms. But lead author Gad Asher, a senior scientist at the Weizmann Institute of Science in Rehovot, Israel, and his colleagues, including postdoctoral fellows Yaarit Adamovich and Benjamin Ladeuix, wondered if oxygen might also cue circadian rhythms since oxygen absorption in animals varies alongside meals and changing temperatures.
In the paper, the researchers show that changing the concentration of oxygen in cells by just 3%, twice a day, will synchronize mouse cells to a circadian rhythm. They suspected HIF1α was the link between oxygen and the circadian clock because HIF1α plays both a role in oxygen homeostasis in cells. They found that cells with low HIF1α levels won’t synchronize in response to oxygen variations.
“It was extremely exciting to see that even small changes in oxygen levels were sufficient to efficiently reset the circadian clock,” says Asher. “The study actually raises a lot of important questions; although we show that clock reset by oxygen is dependent on HIF1α, we did not yet fully identify how HIF1α integrates within the core clock circuitry.”
The researchers further explored oxygen’s effect on circadian rhythms with jetlag experiments. Just like humans, mice are prone to jetlag after a sudden shift in daylight hours. Mice were first left to eat, sleep and run on their wheels in air-controlled environments. Altering oxygen levels alone did not change their circadian rhythms but once mice experienced a 6-hour jump ahead in daylight hours, varying oxygen levels could help them adapt their eating, sleeping and running habits to the new time faster. They also saw that a small drop in oxygen levels 12 hours before the 6-hour daylight shift, or 2 hours afterwards, put the mice back on their circadian schedules faster and this too was dependent on HIF1α levels.
Presently, commercial airliners pressurize cabins to the same air density of a city 6,000-8,000 feet above sea level. This low-pressure saves wear and tear on the airplane, but enough passengers suffer from airsickness in response to this drop in oxygen levels that some airlines are considering ways to increase the pressure on flights. In fact, Boeing designed its new 787 Dreamliner so that it can be pressurized to the equivalent of lower altitudes for this reason. But in light of these findings, the researchers noted passengers may feel better with higher pressurized cabins during flights, but may also lose a potential advantage of recovering from jetlag. And in light of the effects of lower oxygen levels, the researchers now want to see what higher oxygen levels may do to the circadian clock.
“We are very looking forward to seeing the outcome of these experiments – it will be interesting both from basic science and also from a practical standpoint,” said Asher. “I believe passengers might be more enthusiastic to inhale oxygen-enriched air to alleviate jetlag in contrast to low oxygen.”
Understanding how oxygen influences the circadian clock goes beyond jetlag. Cardiovascular disease, COPD, shift work sleep disorder, and other common health problems can result in tissues with low oxygen levels. “We show that oxygen works in mammals, specifically rodents, but it will be interesting to test whether oxygen can reset the clock of bacteria, plants, flies and additional organisms,” says Asher.
Funding: This work was supported by the Israel Science Foundation, the European Research Council, the Yeda Sela Center for Basic Research, the British Heart Foundation and the European Union, Seventh Framework Programme, Marie Curie Actions.
Source: Joseph Caputo – Cell Press
Image Source: NeuroscienceNews.com image is credited to Asher et al./Cell Metabolism 2016.
Original Research: Full open access research for “Rhythmic Oxygen Levels Reset Circadian Clocks through HIF1α” by Yaarit Adamovich, Benjamin Ladeuix, Marina Golik, Maarten P. Koeners, and Gad Asher in Cell Metabolism. Published online October 20 2016 doi:10.1016/j.cmet.2016.09.014
[cbtabs][cbtab title=”MLA”]Cell Press “Blast of Thin Air Can Reset Circadian Clock.” NeuroscienceNews. NeuroscienceNews, 17 October 2016.
<https://neurosciencenews.com/circadian-clock-oxygen-5321/>.[/cbtab][cbtab title=”APA”]Cell Press (2016, October 17). Blast of Thin Air Can Reset Circadian Clock. NeuroscienceNew. Retrieved October 17, 2016 from https://neurosciencenews.com/circadian-clock-oxygen-5321/[/cbtab][cbtab title=”Chicago”]Cell Press “Blast of Thin Air Can Reset Circadian Clock.” https://neurosciencenews.com/circadian-clock-oxygen-5321/ (accessed October 17, 2016).[/cbtab][/cbtabs]
Abstract
Rhythmic Oxygen Levels Reset Circadian Clocks through HIF1α
Highlights
•O2 levels exhibit daily rhythms in blood and tissue of rodents
•Physiological O2 rhythms reset clocks in cultured cells in HIF1α-dependent manner
•Several core clock genes respond to changes in O2 levels in HIF1α-dependent fashion
•Modulation of O2 levels accelerates the recovery of mice in a jet lag protocol
Summary
The mammalian circadian system consists of a master clock in the brain that synchronizes subsidiary oscillators in peripheral tissues. The master clock maintains phase coherence in peripheral cells through systemic cues such as feeding-fasting and temperature cycles. Here, we examined the role of oxygen as a resetting cue for circadian clocks. We continuously measured oxygen levels in living animals and detected daily rhythms in tissue oxygenation. Oxygen cycles, within the physiological range, were sufficient to synchronize cellular clocks in a HIF1α-dependent manner. Furthermore, several clock genes responded to changes in oxygen levels through HIF1α. Finally, we found that a moderate reduction in oxygen levels for a short period accelerates the adaptation of wild-type but not of HIF1α-deficient mice to the new time in a jet lag protocol. We conclude that oxygen, via HIF1α activation, is a resetting cue for circadian clocks and propose oxygen modulation as therapy for jet lag.
“Rhythmic Oxygen Levels Reset Circadian Clocks through HIF1α” by Yaarit Adamovich, Benjamin Ladeuix, Marina Golik, Maarten P. Koeners, and Gad Asher in Cell Metabolism. Published online October 20 2016 doi:10.1016/j.cmet.2016.09.014






