Summary: A new study provides a description of metals in the brain that may drive the progression of Alzheimer’s disease.
Source: University of Warwick.
Alzheimer’s disease could be better treated, thanks to a breakthrough discovery of the properties of the metals in the brain involved in the progression of the neurodegenerative condition, by an international research collaboration including the University of Warwick.
Dr Joanna Collingwood, from Warwick’s School of Engineering, was part of a research team which characterised iron species associated with the formation of amyloid protein plaques in the human brain – abnormal clusters of proteins in the brain. The formation of these plaques is associated with toxicity which causes cell and tissue death, leading to mental deterioration in Alzheimer’s patients.
They found that in brains affected by Alzheimer’s, several chemically-reduced iron species including a proliferation of a magnetic iron oxide called magnetite – which is not commonly found in the human brain – occur in the amyloid protein plaques. The team had previously shown that these minerals can form when iron and the amyloid protein interact with each other. Thanks to advanced measurement capabilities at synchrotron X-ray facilities in the UK and USA, including the Diamond Light Source I08 beamline in Oxfordshire, the team has now shown detailed evidence that these processes took place in the brains of individuals who had Alzheimer’s disease. They also made unique observations about the forms of calcium minerals present in the amyloid plaques.
Understanding the significance of these metals to the progression of Alzheimer’s could lead to more effective future therapies which combat the disease at its root.
Dr Joanna Collingwood, Associate Professor at the University of Warwick’s School of Engineering and expert in trace metals analysis, high resolution imaging, and neurodegenerative disorders, commented:
“Iron is an essential element in the brain, so it is critical to understand how its management is affected in Alzheimer’s disease. The advanced X-ray techniques that we used in this study have delivered a step-change in the level of information that we can obtain about iron chemistry in the amyloid plaques. We are excited to have these new insights into how amyloid plaque formation influences iron chemistry in the human brain, as our findings coincide with efforts by others to treat Alzheimer’s disease with iron-modifying drugs.”
The team, led by an EPSRC-funded collaboration between University of Warwick and Keele University – and which includes researchers from University of Florida and The University of Texas at San Antonio – made their discovery by extracting amyloid plaque cores from two deceased patients who had a formal diagnosis of Alzheimer’s.
The researchers scanned the plaque cores using state-of-the-art X-ray microscopy at the Advanced Light Source in Berkeley, USA and at beamline I08 at the Diamond Light Source synchrotron in Oxfordshire, to determine the chemical properties of the minerals within them.
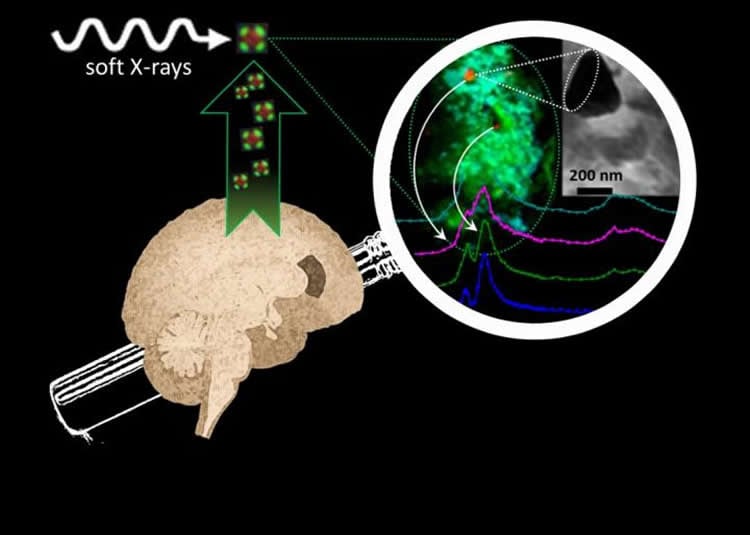
They also analysed the magnetic state of the iron species in the plaques to confirm the presence of various iron minerals including the magnetic iron oxide magnetite.
The research team propose that interactions between iron and amyloid that produce the chemically reduced iron species, including magnetite, may account for toxicity that contributes to the development and progression of Alzheimer’s.
There are 850,000 people with dementia in the UK, with numbers set to rise to over 1 million by 2025. This will soar to 2 million by 2051.
There is no cure for Alzheimer’s disease or any other type of dementia. Delaying the onset of dementia by five years would halve the number of deaths from the condition, saving 30,000 lives a year.
Funding: This work was supported by EPSRC grants EP/K035193/1 (JFC), EP/N033191/1-EP/N033140/1 (JFC-NDT), the Alzheimer’s Association (AARFD-17-529742), University of Warwick alumni donations (VTT, JE), the RCMI Program from NIH at UTSA (5G12RR013646, G12MD007591), San Antonio Life Sciences Institute (SALSI)-Clusters in Research Excellence Program, and Semmes Foundation.
Source: Tom Frew – University of Warwick
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to University of Warwick/Dr Joanna Collingwood.
Original Research: Abstract for “Nanoscale synchrotron X-ray speciation of iron and calcium compounds in amyloid plaque cores from Alzheimer’s disease subjects” by James Everett, Joanna F. Collingwood, Vindy Tjendana-Tjhin, Jake Brooks, Frederik Lermyte, Germán Plascencia-Villa, Ian Hands-Portman, Jon Dobson, George Perry and Neil D. Telling in Nanoscale. Published June 21 2018.
doi:10.1039/C7NR06794A
[cbtabs][cbtab title=”MLA”]University of Warwick “Alzheimer’s Breakthrough: Brain Metals that May Drive Disease Progression Revealed.” NeuroscienceNews. NeuroscienceNews, 21 June 2018.
<https://neurosciencenews.com/brain-metal-alzheimers-9411/>.[/cbtab][cbtab title=”APA”]University of Warwick (2018, June 21). Alzheimer’s Breakthrough: Brain Metals that May Drive Disease Progression Revealed. NeuroscienceNews. Retrieved June 21, 2018 from https://neurosciencenews.com/brain-metal-alzheimers-9411/[/cbtab][cbtab title=”Chicago”]University of Warwick “Alzheimer’s Breakthrough: Brain Metals that May Drive Disease Progression Revealed.” https://neurosciencenews.com/brain-metal-alzheimers-9411/ (accessed June 21, 2018).[/cbtab][/cbtabs]
Abstract
Nanoscale synchrotron X-ray speciation of iron and calcium compounds in amyloid plaque cores from Alzheimer’s disease subjects
Altered metabolism of biometals in the brain is a key feature of Alzheimer’s disease, and biometal interactions with amyloid-β are linked to amyloid plaque formation. Iron-rich aggregates, including evidence for the mixed-valence iron oxide magnetite, are associated with amyloid plaques. To test the hypothesis that increased chemical reduction of iron, as observed in vitro in the presence of aggregating amyloid-β, may occur at sites of amyloid plaque formation in the human brain, the nanoscale distribution and physicochemical states of biometals, particularly iron, were characterised in isolated amyloid plaque cores from human Alzheimer’s disease cases using synchrotron X-ray spectromicroscopy. In situ X-ray magnetic circular dichroism revealed the presence of magnetite: a finding supported by ptychographic observation of an iron oxide crystal with the morphology of biogenic magnetite. The exceptional sensitivity and specificity of X-ray spectromicroscopy, combining chemical and magnetic probes, allowed enhanced differentiation of the iron oxides phases present. This facilitated the discovery and speciation of ferrous-rich phases and lower oxidation state phases resembling zero-valent iron as well as magnetite. Sequestered calcium was discovered in two distinct mineral forms suggesting a dynamic process of amyloid plaque calcification in vivo. The range of iron oxidation states present and the direct observation of biogenic magnetite provide unparalleled support for the hypothesis that chemical reduction of iron arises in conjunction with the formation of amyloid plaques. These new findings raise challenging questions about the relative impacts of amyloid-β aggregation, plaque formation, and disrupted metal homeostasis on the oxidative burden observed in Alzheimer’s disease.







