Research led by UCL, Yale and University of California, San Francisco has shown that the hormone estrogen alleviates the sleep disruption experienced by zebrafish genetically designed to help understand the biology of autism spectrum disorder (ASD).
The scientists set out to investigate the function of genes linked to autism and seizures in humans by using zebrafish as a model system. They unexpectedly found that estrogens have a selective effect in calming hyperactive fish during the night which will help scientists to understand the brain pathways affected in ASD. The finding is also intriguing given ASD is four times more common in men than women.
Co-corresponding author, Dr Jason Rihel, UCL Cell & Developmental Biology, said: “We’re surprised to see that estrogens in particular have such a selective effect in correcting hyperactive behaviour in our ASD fish model, and we’re investigating this further to understand the mechanisms of this response. We don’t know if the hormone is targeting single or multiple pathways, as estrogen is involved in lots of processes, but we’re keen to find out.”
The team discovered that a plant-derived estrogen, called biochanin A, and the human sex hormone, β-estradiol, were most effective at selectively stopping night time hyperactivity without affecting the daytime activity of the fish. The FDA approved drug risperidone, which was the first approved to treat irritability and aggressive behaviour in ASD patients, stopped hyperactivity but made fish less active during the day, suggesting estrogens are better at selectively targeting pathways that are relevant to ASD.
First author, Dr Ellen Hoffman, Yale University, said: “This research helps scientists to understand the function of an autism risk gene in the developing brain, which is important for understanding the biology of autism. The mechanism of action of the estrogens on zebrafish behaviour remains unknown and there is considerably more work to do before these findings can be applied to humans.”
The study, published today in Neuron, used a simple animal model with genetic mutations to understand ASD at different levels, from cellular processes and brain circuitry, through to behaviour. Zebrafish were chosen as they have complex behaviours like sleep at a young age and are transparent, allowing brain development and activity to be visualised with markers. They are also amenable to drug screening, as compounds added to the water will enter the larval zebrafish brain.
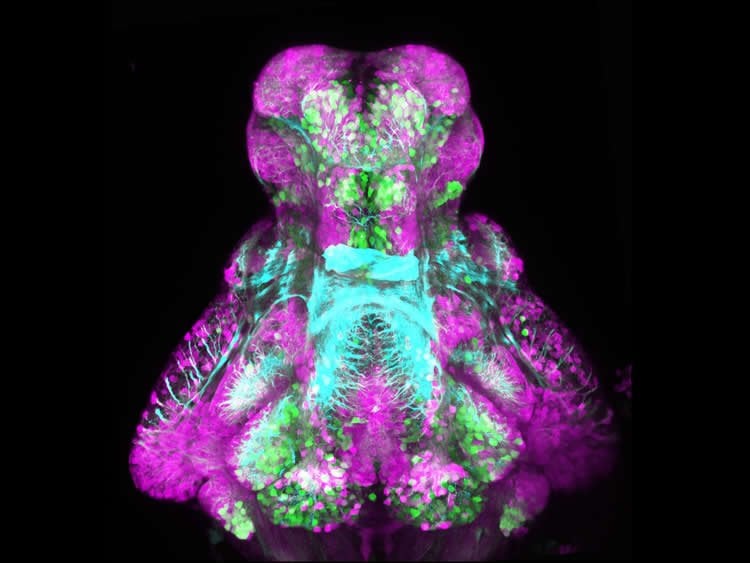
Normal zebrafish were studied alongside those predisposed to seizures and sleep disruption due a mutation in the CNTNAP2 (often pronounced ‘catnap’) gene, which has been associated with human ASD. To identify specific processes involved in ASD-linked hyperactivity, the behavioural patterns of the mutant fish were compared to normal zebrafish exposed to 550 psychoactive compounds. The compounds predicted to trigger or suppress the abnormal behaviour became candidates for follow-up experiments.
Dr Rihel added, “We used the drug screen as a tool to help identify the core mechanisms involved in ASD rather than to find new therapies. For example, drugs predicted to cause sleeplessness in the normal fish population might indicate the same pathways are affected in the mutant population. Conversely, those drugs that selectively promote sleep may hint at ways to rescue the mutant population from hyperactivity.” Four of the top 10 compounds the screen predicted to supress sleeplessness were known to have estrogenic activity, suggesting estrogens as a whole class of molecules may have a positive effect on the mutant fish population. This was tested on both normal and mutant fish siblings using different doses of biochanin A and β-estradiol, which showed that both compounds selectively suppress the mutant night time hyperactivity.
The team plan to use this information to test the effect of estrogens in fish with other genetic mutations linked to ASD before progressing to mammalian models. These further studies will help map the pathways involved in ASD and understand the mechanism of action by which estrogens can specifically alter behaviour.
Funding: The UCL contribution to the study was funded by the Biotechnology and Biological Sciences Research Council (BBSRC), the European Research Council, the Wellcome Trust and UCL.
Source: Bex Caygill – UCL
Image Source: The image is credited to Kate Turner, UCL
Original Research: Abstract for “Estrogens Suppress a Behavioral Phenotype in Zebrafish Mutants of the Autism Risk Gene, CNTNAP2” by Ellen J. Hoffman, Katherine J. Turner, Joseph M. Fernandez, Daniel Cifuentes, Marcus Ghosh, Sundas Ijaz, Roshan A. Jain, Fumi Kubo, Brent R. Bill, Herwig Baier, Michael Granato, Michael J.F. Barresi, Stephen W. Wilson, Jason Rihel, Matthew W. State, and Antonio J. Giraldez in Neuron. Published online December 21 2015 doi:10.1016/j.neuron.2015.12.039
Abstract
Estrogens Suppress a Behavioral Phenotype in Zebrafish Mutants of the Autism Risk Gene, CNTNAP2
Highlights
•Zebrafish mutants of the autism risk gene cntnap2 have GABAergic neuron deficits
•High-throughput behavioral profiling identifies nighttime hyperactivity in mutants
•cntnap2 mutants exhibit altered responses to GABAergic and glutamatergic compounds
•Estrogenic compounds suppress the cntnap2 mutant behavioral phenotype
Summary
Autism spectrum disorders (ASDs) are a group of devastating neurodevelopmental syndromes that affect up to 1 in 68 children. Despite advances in the identification of ASD risk genes, the mechanisms underlying ASDs remain unknown. Homozygous loss-of-function mutations in Contactin Associated Protein-like 2 (CNTNAP2) are strongly linked to ASDs. Here we investigate the function of Cntnap2 and undertake pharmacological screens to identify phenotypic suppressors. We find that zebrafish cntnap2 mutants display GABAergic deficits, particularly in the forebrain, and sensitivity to drug-induced seizures. High-throughput behavioral profiling identifies nighttime hyperactivity in cntnap2 mutants, while pharmacological testing reveals dysregulation of GABAergic and glutamatergic systems. Finally, we find that estrogen receptor agonists elicit a behavioral fingerprint anti-correlative to that of cntnap2 mutants and show that the phytoestrogen biochanin A specifically reverses the mutant behavioral phenotype. These results identify estrogenic compounds as phenotypic suppressors and illuminate novel pharmacological pathways with relevance to autism.
“Estrogens Suppress a Behavioral Phenotype in Zebrafish Mutants of the Autism Risk Gene, CNTNAP2” by Ellen J. Hoffman, Katherine J. Turner, Joseph M. Fernandez, Daniel Cifuentes, Marcus Ghosh, Sundas Ijaz, Roshan A. Jain, Fumi Kubo, Brent R. Bill, Herwig Baier, Michael Granato, Michael J.F. Barresi, Stephen W. Wilson, Jason Rihel, Matthew W. State, and Antonio J. Giraldez in Neuron. Published online December 21 2015 doi:10.1016/j.neuron.2015.12.039






