Summary: A deficiency of the SHANK3 gene, a gene associated with ASD, results in structural and functional deficits in the prefrontal cortex. The functional and structural alterations in the PFC were linked to an impairment in social interaction in male mice.
Source: SfN
Mice lacking the gene Shank3 display structural and functional deficits in the prefrontal cortex finds a study published in Journal of Neuroscience. The research advances our understanding of one of the most common genetic risk factors for autism spectrum disorders.
Shank3 mutations are commonly observed in humans with autism and related developmental disorders. Previous animal research documents associations between Shank3 and basal ganglia dysfunction, which may contribute to repetitive behaviors characteristic of autism. In humans, Shank3 mutations have also been linked to intellectual and language difficulties.
Using magnetic resonance imaging in adult male mice, Marco Pagani, Alessandro Gozzi and colleagues demonstrate reduced prefrontal cortex connectivity and gray matter volume in Shank3-deficient animals.
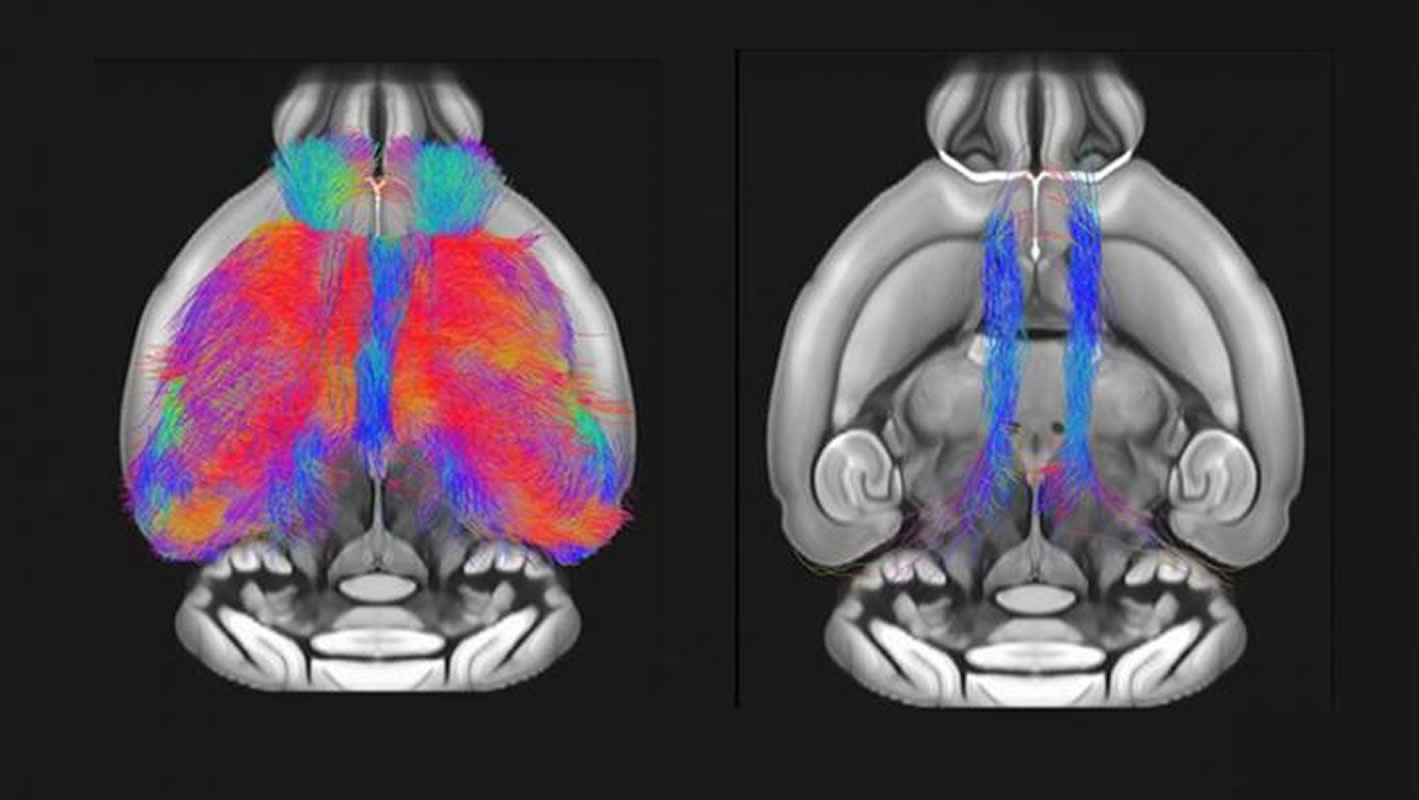
The researchers report these brain differences are tightly linked to impaired social interaction with male mice.
These findings establish a role of Shank3 in maintaining prefrontal cortex connectivity, which may increase autism risk when disrupted.
Source:
SfN
Media Contacts:
David Barnstone – SfN
Image Source:
The image is credited to Pagani et al., JNeurosci (2019).
Original Research: Closed access
“Deletion of autism risk gene Shank3 disrupts prefrontal connectivity”. Marco Pagani, Alice Bertero, Adam Liska, Alberto Galbusera, Mara Sabbioni, Noemi Barsotti, Nigel Colenbier, Daniele Marinazzo, Maria Luisa Scattoni, Massimo Pasqualetti and Alessandro Gozzi.
Journal of Neuroscience doi:10.1523/JNEUROSCI.2529-18.2019
Abstract
Deletion of autism risk gene Shank3 disrupts prefrontal connectivity
Mutations in the synaptic scaffolding protein Shank3 are a major cause of autism, and are associated with prominent intellectual and language deficits. However, the neural mechanisms whereby SHANK3 deficiency affects higher order socio-communicative functions remain unclear. Using high-resolution functional and structural MRI in adult male mice, here we show that loss of Shank3 (Shank3B-/-) results in disrupted local and long-range prefrontal and fronto-striatal functional connectivity. We document that prefrontal hypo-connectivity is associated with reduced short-range cortical projections density, and reduced gray matter volume. Finally, we show that prefrontal disconnectivity is predictive of social communication deficits, as assessed with ultrasound vocalization recordings. Collectively, our results reveal a critical role of SHANK3 in the development of prefrontal anatomy and function, and suggest that SHANK3 deficiency may predispose to intellectual disability and socio-communicative impairments via dysregulation of higher-order cortical connectivity.
Significance
Mutations in the synaptic scaffolding protein SHANK3 are commonly associated with autism, intellectual and language deficits. Previous research has linked SHANK3 deficiency to basal ganglia dysfunction, motor stereotypies and social deficits. However, the neural mechanism whereby Shank3 gene mutations affects cortical functional connectivity and higher order socio-communicative functions remain unclear. Here we show that loss of SHANK3 in mice results in largely disrupted functional connectivity and abnormal gray matter anatomy in prefrontal areas. We also show that prefrontal connectivity disruption is tightly linked to socio-communicative deficits. Our findings suggest that SHANK3 is a critical orchestrator of fronto-cortical function, and that disrupted connectivity of prefrontal areas may underpin socio-communicative impairments observed in SHANK3 mutation carriers.






