Summary: Investigating the effects of genetic abnormalities associated with autism and human brain development, researchers found brain organoids engineered to have lower levels of the ASD-associated SHANK3 gene had distinct features including neural firing hyperactivity, disruptions in pathways that cause cells to adhere to each other, and indications of ineffective neurotransmission.
Source: University of Utah
Whatever you do, don’t call them “mini-brains,” say University of Utah Health scientists. Regardless, the seed-sized organoids—which are grown in the lab from human cells—provide insights into the brain and uncover differences that may contribute to autism in some people.
“We used to think it would be too difficult to model the organization of cells in the brain,” says Alex Shcheglovitov, PhD, assistant professor of neurobiology at U of U Health. “But these organoids self-organize. Within a few months, we see layers of cells that are reminiscent of the cerebral cortex in the human brain.”
The research describing the organoids and their potential for understanding neural diseases publishes in Nature Communications on Oct 6 with Shcheglovitov as senior author and Yueqi Wang, PhD, a former graduate student in his lab, as lead author. They carried out the research with postdoctoral scientist Simone Chiola, PhD, and other collaborators at the University of Utah, Harvard University, University of Milan, and Montana State University.
Investigating autism
Having the ability to model aspects of the brain in this way gives scientists a glimpse into the inner workings of a living organ that is otherwise nearly impossible to access. And since the organoids grow in a dish, they can be tested experimentally in ways that a brain cannot.
Shcheglovitov’s team used an innovative process to investigate effects of a genetic abnormality associated with autism spectrum disorder and human brain development. They found that organoids engineered to have lower levels of the gene, called SHANK3, had distinct features.
Even though the autism organoid model appeared normal, some cells did not function properly:
- Neurons were hyperactive, firing more often in response to stimuli,
- Other signs indicated neurons may not efficiently pass along signals to other neurons,
- Specific molecular pathways that cause cells to adhere to one another were disrupted.
These findings are helping to uncover the cellular and molecular causes of symptoms associated with autism, the authors say. They also demonstrate that the lab-grown organoids will be valuable for gaining a better understanding of the brain, how it develops, and what goes wrong during disease.
“One goal is to use brain organoids to test drugs or other interventions to reverse or treat disorders,” says Jan Kubanek, PhD, a co-author on the study and an assistant professor of biomedical engineering at the U.
Building a better brain model
Scientists have long searched for suitable models for the human brain. Lab-grown organoids are not new, but previous versions did not develop in a reproduceable way, making experiments difficult to interpret.
To create an improved model, Shcheglovitov’s team took cues from how the brain develops normally. The researchers prompted human stem cells to become neuroepithelial cells, a specific stem cell type that forms self-organized structures, called neural rosettes, in a dish. Over the course of months, these structures coalesced into spheres and increased in size and complexity at a rate similar to the developing brain in a growing fetus.
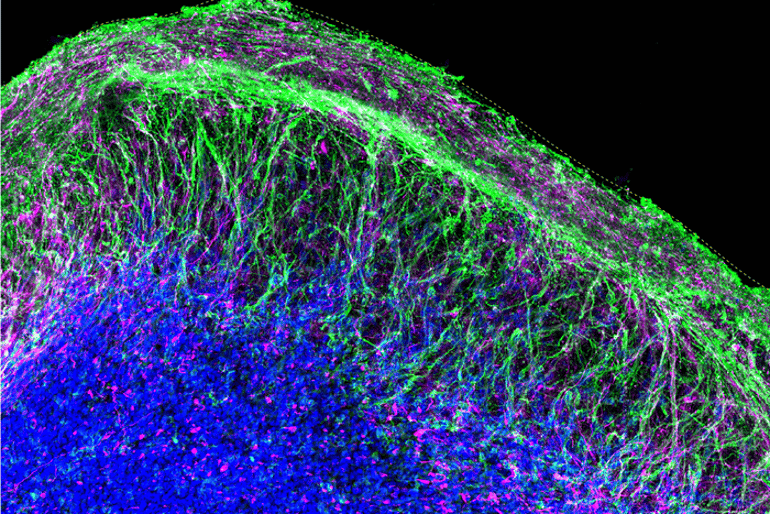
After five months in the lab, the organoids were reminiscent of “one wrinkle of a human brain” at 15 to 19 weeks post-conception, Shcheglovitov says. The structures contained an array of neural and other cell types found in the cerebral cortex, the outermost layer of the brain involved in language, emotion, reasoning, and other high-level mental processes.
Like a human embryo, organoids self-organized in a predictable fashion, forming neural networks that pulsated with oscillatory electrical rhythms and generated diverse electrical signals characteristic of a variety of different kinds of mature brain cells.
“These organoids had patterns of electrophysiological activity that resembled actual activity in the brain. I didn’t expect that,” Kubanek says. “This new approach models most major cell types and in functionally meaningful ways.”
Shcheglovitov explains that these organoids, which more reliably reflect intricate structures in the cortex, will allow scientists to study how specific types of cells in the brain arise and work together to perform more complex functions.
“We’re beginning to understand how complex neural structures in the human brain arise from simple progenitors,” Wang says. “And we’re able to measure disease-related phenotypes using 3D organoids that are derived from stem cells containing genetic mutations.”
He adds that using the organoids, researchers will be able to better investigate what happens at the earliest stages of neurological conditions, before symptoms develop.
Funding: Support for the work came from the National Institutes of Health, Brain Research Foundation, Brain and Behavior Research Foundation, Whitehall Foundation, University of Utah Neuroscience Initiative, and University of Utah Genome Project Initiative.
About this autism research news
Author: Julie Kiefer
Source: University of Utah
Contact: Julie Kiefer – University of Utah
Image: The image is credited to Yueqi Wang
Original Research: Open access.
“Modeling human telencephalic development and autism-associated SHANK3 deficiency using organoids generated from single neural rosettes” by Alex Shcheglovitov et al. Nature Communications
Abstract
Modeling human telencephalic development and autism-associated SHANK3 deficiency using organoids generated from single neural rosettes
Human telencephalon is an evolutionarily advanced brain structure associated with many uniquely human behaviors and disorders. However, cell lineages and molecular pathways implicated in human telencephalic development remain largely unknown.
We produce human telencephalic organoids from stem cell-derived single neural rosettes and investigate telencephalic development under normal and pathological conditions.
We show that single neural rosette-derived organoids contain pallial and subpallial neural progenitors, excitatory and inhibitory neurons, as well as macroglial and periendothelial cells, and exhibit predictable organization and cytoarchitecture.
We comprehensively characterize the properties of neurons in SNR-derived organoids and identify transcriptional programs associated with the specification of excitatory and inhibitory neural lineages from a common pool of NPs early in telencephalic development.
We also demonstrate that neurons in organoids with a hemizygous deletion of an autism- and intellectual disability-associated gene SHANK3 exhibit intrinsic and excitatory synaptic deficits and impaired expression of several clustered protocadherins.
Collectively, this study validates SNR-derived organoids as a reliable model for studying human telencephalic cortico-striatal development and identifies intrinsic, synaptic, and clustered protocadherin expression deficits in human telencephalic tissue with SHANK3 hemizygosity.






