Summary: Researchers report human growth hormone, prepared from human tissue prior to 1985, may have been contaminated with seeds of the amyloid beta protein. In a new study, researchers injected mice with the original c-hGH batches containing amyloid beta which seeded amyloid pathology, even decades after storage. Findings support the hypothesis that the Alzheimer’s causing proteins can be transmitted to from contaminated materials.
Source: UCL.
A UCL-led study has confirmed that some vials of a hormone used in discontinued medical treatments contained seeds of a protein implicated in Alzheimer’s disease, and are able to seed amyloid pathology in mice.
The research, published in Nature, follows on from the team’s 2015 study that found evidence of amyloid pathology in people who had developed Creutzfeldt-Jakob disease (CJD) following treatments with human growth hormone extracted from large pools of pituitary glands removed from deceased individuals at autopsy.*
Amyloid pathology involves a build-up of brain proteins known to cause Alzheimer’s disease.
This new research confirms that certain batches of this hormone did indeed contain seeds of the amyloid beta protein and found, in a new experimental study, that the hormone batches transmitted amyloid pathology to laboratory mice.
The findings support the team’s hypothesis that amyloid beta was accidentally transmitted to patients via this long since discontinued medical treatment.
“Our earlier study found that some individuals who developed CJD many years after treatment as children with cadaver-derived human pituitary growth hormone also had deposits in the brain of a type of aberrant protein characteristic of Alzheimer’s disease,” said the study’s lead author, Professor John Collinge (MRC Prion Unit and UCL Institute of Prion Diseases).
“We suggested that the human growth hormone – which prior to 1985 was prepared from human tissue – may have been contaminated with seeds of this material as well as the prions causing CJD. Our latest findings now confirm that some archived batches of this hormone did indeed contain seeds of the amyloid beta protein found in Alzheimer’s disease.
“It is most important to emphasise that there is no suggestion in our work that you can catch Alzheimer’s disease (or indeed CJD) by contact with a person with the disease. Our findings highlight the need to do further research in this area.”
The 2015 findings linked childhood growth hormone injections transfusions to amyloid pathology in adults, by studying autopsied brains from eight people who had died from CJD.
All eight people whose cases were described in the 2015 paper had previously been treated with cadaveric pituitary-derived human growth hormone (c-hGH), which up until 1985 had been used to treat various causes of short stature before concerns were raised about transmission of the prions (seeds of misshapen prion protein) that cause CJD. The use of cadaveric c-hGH was then discontinued and replaced with synthetic hormone that did not carry the risk of transmitting CJD.
Six of the people who died were found to have amyloid pathology in brain tissue, four of whom also had some degree of cerebral amyloid angiopathy (CAA), a disease involving the build-up of amyloid beta in blood vessels in the brain (which can cause bleeding in the brain). CAA is also seen to some degree in most patients with Alzheimer’s disease. None of these CJD patients had developed the full picture of Alzheimer’s disease at the time of their death from CJD.
For the current study, the research team tracked down some of the batches of c-hGH that the patients had been treated with and tested them, finding that some of the samples did in fact have significant levels of amyloid beta proteins (implicated in both CAA and Alzheimer’s) and tau proteins (implicated in Alzheimer’s disease).
A diagnosis of Alzheimer’s disease requires detection of both aberrant proteins, amyloid beta and tau.
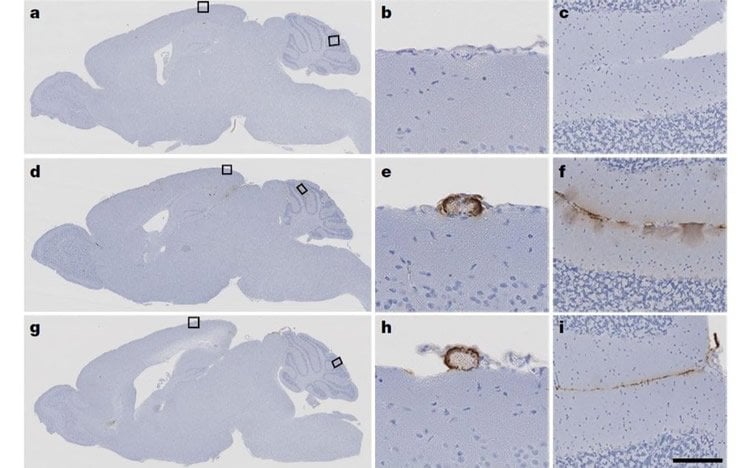
The researchers then tested whether this material was capable of seeding pathology by injecting samples from the hormone vials into laboratory mice that had been genetically modified to be prone to the amyloid beta pathology seen in Alzheimer’s disease (the mice used in the study were not prone to tau).
The mice that had been inoculated with the contaminated growth hormone samples showed clear seeding of amyloid pathology in their brains and developed CAA within less than a year, just like mice that had been injected with tissue from patients with typical Alzheimer’s disease in another part of this study. Groups of the same mice that had been injected with synthetic growth hormone or normal brain tissue as controls showed no such patterns.
The findings demonstrate that the original c-hGH batches contained amyloid beta proteins that can seed amyloid pathology in mice, even after decades of storage.
“We have now provided experimental evidence to support our hypothesis that amyloid beta pathology can be transmitted to people from contaminated materials,” said Professor Collinge.
“We cannot yet confirm whether medical or surgical procedures have ever caused Alzheimer’s disease itself in people, or how common it might be to acquire amyloid pathology in this way.”
“It will be important to review risks of transmission of amyloid pathology by other medical procedures still done today, including instruments used in brain surgery, drawing on other research and what we already know about accidental CJD transmission,”** he added.
“While we are concerned about what this could mean about how amyloid pathology might be transmitted, we also aim to understand more about the cause and consequences of amyloid beta deposition in the brain and its relationship to tau aggregation, the other hallmark for Alzheimer’s disease,” said co-author Dr Silvia Purro (UCL Institute of Prion Diseases).
Dr Rob Buckle, Chief Science Officer at the Medical Research Council, which funded the study, said: “This study provides new insights into the molecular mechanisms underlying the role of amyloid in Alzheimer’s disease. However, these experiments were in mice predisposed to develop Alzheimer’s disease pathology and there is currently no evidence that AD can be transmitted between people.”
Funding: The study was principally funded by the Medical Research Council with support from the National Institute for Health Research, and involved researchers at the MRC Prion Unit at UCL, UCL Institute of Prion Diseases; Brigham and Women’s Hospital and Harvard Medical School; and RIKEN Center for Brain Science, Japan.
Source: Chris Lane – UCL
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Purro et al, 2018.
Original Research: Abstract for “Transmission of amyloid-β protein pathology from cadaveric pituitary growth hormone” by Silvia A. Purro, Mark A. Farrow, Jacqueline Linehan, Tamsin Nazari, David X. Thomas, Zhicheng Chen, David Mengel, Takashi Saito, Takaomi Saido, Peter Rudge, Sebastian Brandner, Dominic M. Walsh & John Collinge in Nature. Published December 13 2018.
doi:10.1038/s41586-018-0790-y
[cbtabs][cbtab title=”MLA”]UCL”Alzheimer’s Protein Accidentally Transmitted to Patients Via Discontinued Medical Treatments.” NeuroscienceNews. NeuroscienceNews, 14 December 2018.
<https://neurosciencenews.com/amyloid-beta-transmission-10347/>.[/cbtab][cbtab title=”APA”]UCL(2018, December 14). Alzheimer’s Protein Accidentally Transmitted to Patients Via Discontinued Medical Treatments. NeuroscienceNews. Retrieved December 14, 2018 from https://neurosciencenews.com/amyloid-beta-transmission-10347/[/cbtab][cbtab title=”Chicago”]UCL”Alzheimer’s Protein Accidentally Transmitted to Patients Via Discontinued Medical Treatments.” https://neurosciencenews.com/amyloid-beta-transmission-10347/ (accessed December 14, 2018).[/cbtab][/cbtabs]
Abstract
Transmission of amyloid-β protein pathology from cadaveric pituitary growth hormone
We previously reported the presence of amyloid-β protein (Aβ) deposits in individuals with Creutzfeldt–Jakob disease (CJD) who had been treated during childhood with human cadaveric pituitary-derived growth hormone (c-hGH) contaminated with prions. The marked deposition of parenchymal and vascular Aβ in these relatively young individuals with treatment-induced (iatrogenic) CJD (iCJD), in contrast to other prion-disease patients and population controls, allied with the ability of Alzheimer’s disease brain homogenates to seed Aβ deposition in laboratory animals, led us to argue that the implicated c-hGH batches might have been contaminated with Aβ seeds as well as with prions. However, this was necessarily an association, and not an experimental, study in humans and causality could not be concluded. Given the public health importance of our hypothesis, we proceeded to identify and biochemically analyse archived vials of c-hGH. Here we show that certain c-hGH batches to which patients with iCJD and Aβ pathology were exposed have substantial levels of Aβ40, Aβ42 and tau proteins, and that this material can seed the formation of Aβ plaques and cerebral Aβ−amyloid angiopathy in intracerebrally inoculated mice expressing a mutant, humanized amyloid precursor protein. These results confirm the presence of Aβ seeds in archived c-hGH vials and are consistent with the hypothesized iatrogenic human transmission of Aβ pathology. This experimental confirmation has implications for both the prevention and the treatment of Alzheimer’s disease, and should prompt a review of the risk of iatrogenic transmission of Aβ seeds by medical and surgical procedures long recognized to pose a risk of accidental prion transmission.






