Why do some people remain healthy into their 80s and beyond, while others age faster and suffer serious diseases decades earlier? New research led by UCLA life scientists may produce a new way to answer that question — and an approach that could help delay declines in health.
Specifically, the study suggests that analyzing intestinal bacteria could be a promising way to predict health outcomes as we age.
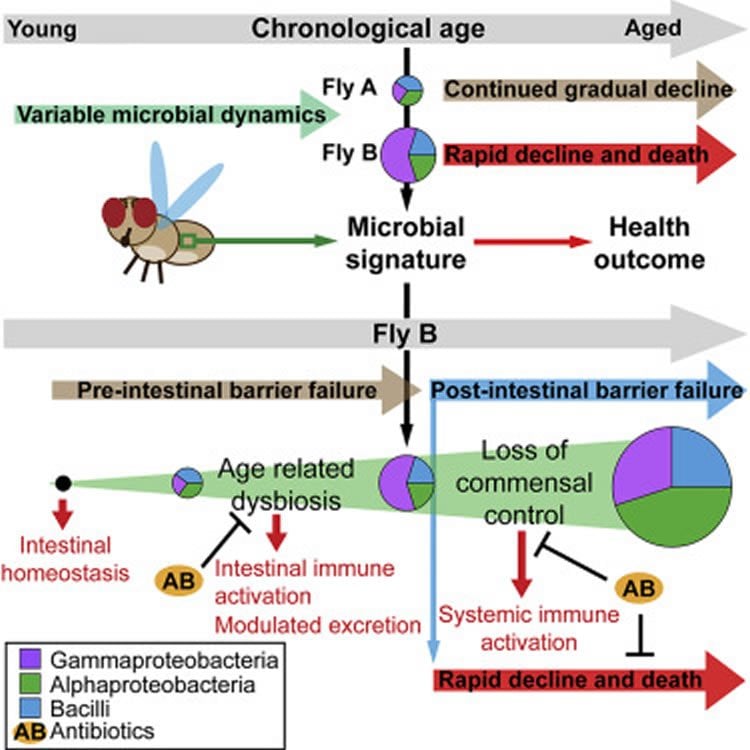
The researchers discovered changes within intestinal microbes that precede and predict the death of fruit flies. The findings were published in the open-source journal Cell Reports.
“Age-onset decline is very tightly linked to changes within the community of gut microbes,” said David Walker, a UCLA professor of integrative biology and physiology, and senior author of the research. “With age, the number of bacterial cells increase substantially and the composition of bacterial groups changes.”
The study used fruit flies in part because although their typical life span is just eight weeks, some live to the age equivalent of humans’ 80s and 90s, while others age and die much younger. In addition, scientists have identified all of the fruit fly’s genes and know how to switch individual ones on and off.
In a previous study, the UCLA researchers discovered that five or six days before flies died, their intestinal tracts became more permeable and started leaking.
In the latest research, which analyzed more than 10,000 female flies, the scientists found that they were able to detect bacterial changes in the intestine before the leaking began. As part of the study, some fruit flies were given antibiotics that significantly reduce bacterial levels in the intestine; the study found that the antibiotics prevented the age-related increase in bacteria levels and improved intestinal function during aging.
The biologists also showed that reducing bacterial levels in old flies can significantly prolong their life span.
“When we prevented the changes in the intestinal microbiota that were linked to the flies’ imminent death by feeding them antibiotics, we dramatically extended their lives and improved their health,” Walker said. (Microbiota are the bacteria and other microorganisms that are abundant in humans, other mammals, fruit flies and many other animals.)
Flies with leaky intestines that were given antibiotics lived an average of 20 days after the leaking began — a substantial part of the animal’s life span. On average, flies with leaky intestines that did not receive antibiotics died within a week.
The intestine acts as a barrier to protect our organs and tissue from environmental damage.
“The health of the intestine — in particular the maintenance of the barrier protecting the rest of the body from the contents of the gut — is very important and might break down with aging,” said Rebecca Clark, the study’s lead author. Clark was a UCLA postdoctoral scholar when the research was conducted and is now a lecturer at England’s Durham University.
The biologists collaborated with William Ja, an assistant professor at Florida’s Scripps Research Institute, and Ryuichi Yamada, a postdoctoral research associate in Ja’s laboratory, to produce an additional group of flies that were completely germ-free, with no intestinal microbes. Those flies showed a very dramatic delay in intestinal damage, and they lived for about 80 days, approximately one-and-a-half times as long as the animal’s typical life span.
Scientists have recently begun to connect a wide variety of diseases, including diabetes and Parkinson’s, among many others, to changes in the microbiota, but they do not yet know exactly what healthy microbiota look like.
“One of the big questions in the biology of aging relates to the large variation in how we age and how long we live,” said Walker, who added that scientific interest in intestinal microbes has exploded in the last five years.
When a fruit fly’s intestine begins to leak, its immune response increases substantially and chronically throughout its body. Chronic immune activation is linked with age-related diseases in people as well, Walker said.
Walker said that the study could lead to realistic ways for scientists to intervene in the aging process and delay the onset of Parkinson’s disease, Alzheimer’s disease, cancer, stroke, cardiovascular disease, diabetes and other diseases of aging — although such progress could take many years, he said.
Other co-authors of the research are Matteo Pellegrini, professor of molecular, cell and developmental biology and co-director of UCLA’s Institute for Quantitative and Computational Biosciences, postdoctoral scholars Anna Salazar and Anil Rana, graduate students Jeanette Alcaraz and Marco Morselli, and researcher Sorel Fitz-Gibbon, all of UCLA; and Michael Rera, a former UCLA postdoctoral scholar.
Funding: The research was funded by the National Institutes of Health’s National Institute on Aging (grants R01AG037514, R01AG049157 and R01AG040288) and the UCLA Institute of Genomics and Proteomics.
Source: Stuart Wolpert – UCLA
Image Source: The image is credited to Walker et al./Cell Reports and is licensed CC BY 4.0
Original Research: Full open access research for “Distinct Shifts in Microbiota Composition during Drosophila Aging Impair Intestinal Function and Drive Mortality” by Rebecca I. Clark, Anna Salazar, Ryuichi Yamada, Sorel Fitz-Gibbon, Marco Morselli, Jeanette Alcaraz, Anil Rana, Michael Rera, Matteo Pellegrini, William W. Ja, and David W. Walker in Cell Reports. Published online August 27 2015 doi:10.1016/j.celrep.2015.08.004
Abstract
Distinct Shifts in Microbiota Composition during Drosophila Aging Impair Intestinal Function and Drive Mortality
Highlights
•Age-related dysbiosis in Drosophila is characterized by Gammaproteobacteria expansion
•Dysbiosis predicts age-onset intestinal barrier dysfunction and rapid health decline
•Age-related dysbiosis drives changes in excretory function
•Loss of commensal control following intestinal barrier dysfunction drives mortality
Summary
Alterations in the composition of the intestinal microbiota have been correlated with aging and measures of frailty in the elderly. However, the relationships between microbial dynamics, age-related changes in intestinal physiology, and organismal health remain poorly understood. Here, we show that dysbiosis of the intestinal microbiota, characterized by an expansion of the Gammaproteobacteria, is tightly linked to age-onset intestinal barrier dysfunction in Drosophila. Indeed, alterations in the microbiota precede and predict the onset of intestinal barrier dysfunction in aged flies. Changes in microbial composition occurring prior to intestinal barrier dysfunction contribute to changes in excretory function and immune gene activation in the aging intestine. In addition, we show that a distinct shift in microbiota composition follows intestinal barrier dysfunction, leading to systemic immune activation and organismal death. Our results indicate that alterations in microbiota dynamics could contribute to and also predict varying rates of health decline during aging in mammals.
“Distinct Shifts in Microbiota Composition during Drosophila Aging Impair Intestinal Function and Drive Mortality” by Rebecca I. Clark, Anna Salazar, Ryuichi Yamada, Sorel Fitz-Gibbon, Marco Morselli, Jeanette Alcaraz, Anil Rana, Michael Rera, Matteo Pellegrini, William W. Ja, and David W. Walker in Cell Reports. Published online August 27 2015 doi:10.1016/j.celrep.2015.08.004







