Summary: Researchers characterize a novel neural population within the striatum that appears to be responsible for the interplay between acetylcholine and GABA.
Source: Karolinska Institute
A new study from the Department of Neuroscience at Karolinska Institutet characterizes a novel neuronal population in the basal ganglia, responsible for the interaction between two types of neurotransmitters, GABA and acetylcholine.
The study was recently published in Cell Reports.
The striatum is the main input structure of the basal ganglia, a brain region involved in a variety of sensorimotor functions and reinforcement learning. 99% of striatal neurons are inhibitory GABAergic cells, and the only exception is the population of cholinergic interneurons.
“In previous studies we have showed the interactions between cholinergic interneurons and the dopamine system, and here we focused on the interactions between the cholinergic and GABAergic systems in the striatum,” explains Gilad Silberberg, Professor at the Department of Neuroscience, and main author of the study.
The striatum is strongly modulated by acetylcholine and early treatment for Parkinson’s disease was based on the cholinergic system. Cholinergic interneurons have been shown to change their activity in Parkinson’s disease, Huntington’s disease and in various forms of dyskinesia, all of which are disorders related to striatal function.
“Here we wanted to study how the cholinergic activity shapes striatal activity via nicotine receptors, a specific receptor of acetylcholine,” says Anna Tokarska, Ph.D. student in the Silberberg laboratory and first author of the study.
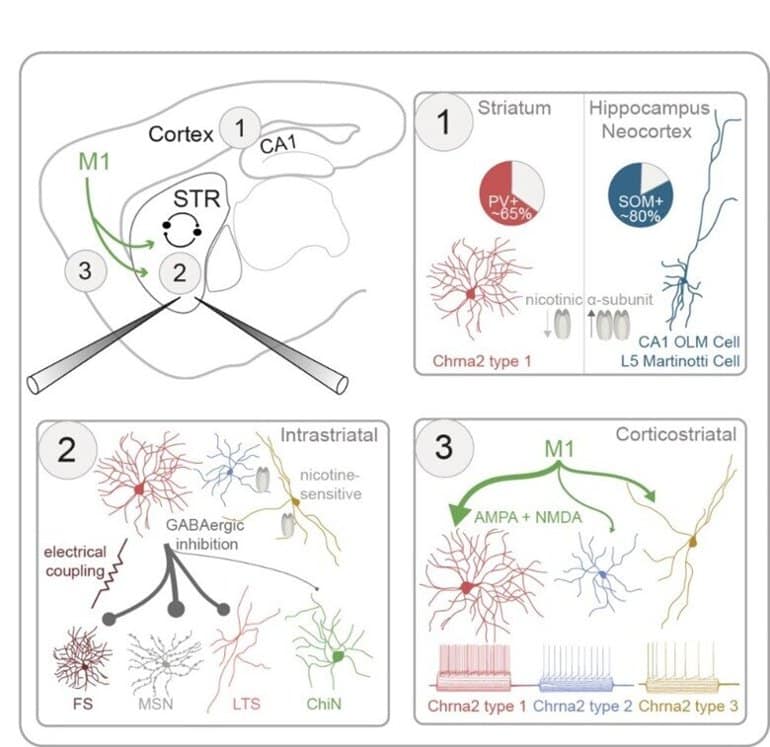
“To do that, we used transgenic mice, marking the striatal interneurons expressing these nicotinic receptors through the Chrna2 gene. We could then use various methods including patch-clamp and optogenetics, to characterize these neurons and their synaptic connectivity in the striatum,” she continues.
The striatal Chrna2 interneuron population was very diverse, including at least three main subpopulations with distinct anatomical and electrical properties. One population was of particular interest, showing novel characteristics including strong response to acetylcholine.
Future steps in this line of research will be to study this population in further detail, including its involvement in striatal function and dysfunction.
About this neuroscience research news
Author: Press Office
Source: Karolinska Institute
Contact: Press Office – Karolinska Institute
Image: The image is credited to Anna Tokarska, Gilad Silberberg
Original Research: Open access.
“GABAergic interneurons expressing the α2 nicotinic receptor subunit are functionally integrated in the striatal microcircuit” by Anna Tokarska et al. Cell Reports
Abstract
GABAergic interneurons expressing the α2 nicotinic receptor subunit are functionally integrated in the striatal microcircuit
Highlights
- Triple whole-cell recordings are used to study striatal interneurons in Chrna2-Cre mice
- Unlike in other brain regions, most striatal Chrna2-interneurons express parvalbumin
- Three distinct subtypes of striatal Chrna2-interneurons are defined
- Their synaptic connectivity is mapped using optogenetics and patch-clamp recordings
Summary
The interactions between the striatal cholinergic and GABAergic systems are crucial in shaping reward-related behavior and reinforcement learning; however, the synaptic pathways mediating them are largely unknown.
Here, we use Chrna2-Cre mice to characterize striatal interneurons (INs) expressing the nicotinic α2 receptor subunit.
Using triple patch-clamp recordings combined with optogenetic stimulations, we characterize the electrophysiological, morphological, and synaptic properties of striatal Chrna2-INs.
Striatal Chrna2-INs have diverse electrophysiological properties, distinct from their counterparts in other brain regions, including the hippocampus and neocortex.
Unlike in other regions, most striatal Chrna2-INs are fast-spiking INs expressing parvalbumin. Striatal Chrna2-INs are intricately integrated in the striatal microcircuit, forming inhibitory synaptic connections with striatal projection neurons and INs, including other Chrna2-INs. They receive excitatory inputs from primary motor cortex mediated by both AMPA and NMDA receptors.
A subpopulation of Chrna2-INs responds to nicotinic input, suggesting reciprocal interactions between this GABAergic interneuron population and striatal cholinergic synapses.






