A new study by Neil Harrison and colleagues published in Biological Psychiatry suggests that a brain reward center, the striatum, may be directly affected by inflammation and that striatal change is related to the emergence of illness behaviors.
Inflammation increases the risk for depression. More specifically, inflammation induces behavioral changes similar to depression that are often associated with illness, including fatigue, difficulty concentrating, lack of motivation, and reduced experience of pleasure.
The authors recruited 23 patients with hepatitis C who were beginning treatment with interferon-alpha (INF-α). This treatment provokes an immediate inflammatory response, confirmed by measuring cytokines in the blood.
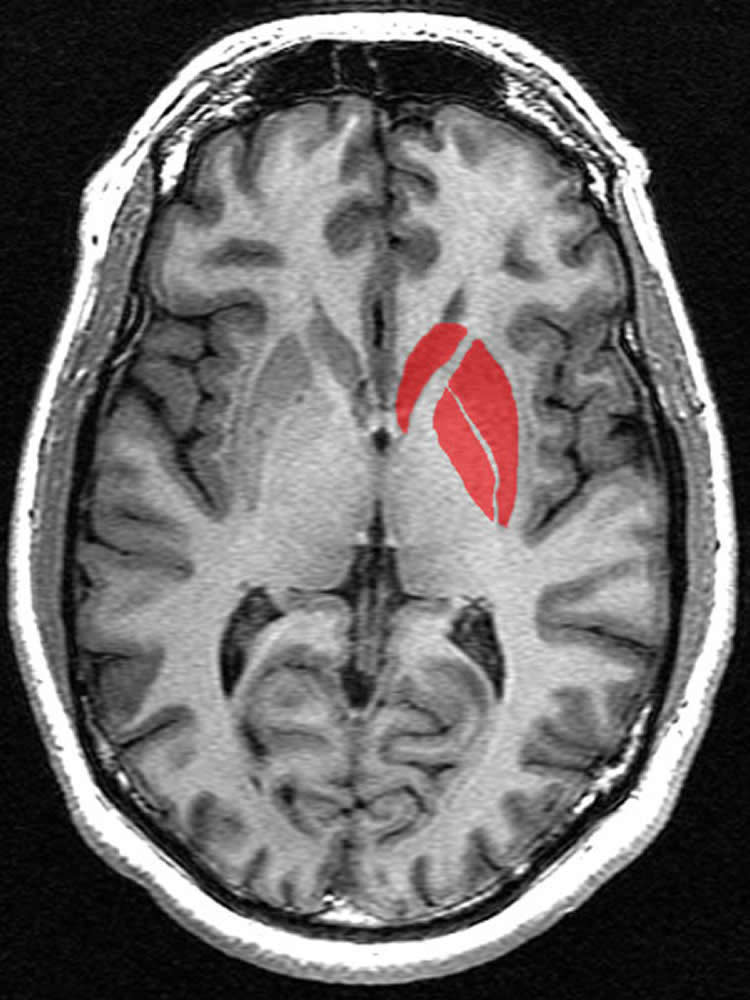
Four hours after INF-α administration, a specialized type of imaging, called magnetization transfer imaging, was performed that showed evidence of microstructural changes in the striatum when compared to scans conducted prior to INF-α administration. This suggests that the striatum is highly sensitive to IFN-α.
IFN-α also induced fatigue and depression in the patients, particularly over weeks 4 through 12 of treatment. Interestingly, the early striatal structural change predicted the later emergence of fatigue, but not depression, in the study participants.
Changes in the striatum were heterogeneous with some changes associated with the risk for fatigue, while other changes seemed to be protective against developing fatigue.
“Inflammation-related fatigue and depression are big clinical problems,” said Dr. John Krystal, Editor of Biological Psychiatry. “This study highlights that the brain regions central to reward and motivation are directly altered by inflammation in ways that that appear to predispose or protect against developing fatigue but not depression. The heterogeneous striatal response may suggest that fatigue and mood are supported by different microcircuits within the striatum.”
“These findings are important as they show that a relatively simple MRI technique can be used to measure effects of inflammation on the brain,” Harrison commented. “Inflammation is increasingly implicated in the cause of common mental illnesses, particularly depression. This technique could be a powerful way to identify patients who are most sensitive to effects of inflammation on the brain. It could also be used to monitor response to novel anti-inflammatory therapies that are now being tested in depression.”
Fudning: Open Access funded by Wellcome Trust.
Source: Rhiannon Bugno – Elsevier
Image Credit: The image is in the public domain
Original Research: Full open access research for “Acute Changes in Striatal Microstructure Predict the Development of Interferon-Alpha Induced Fatigue” by Nicholas G. Dowell, Ella A. Cooper, Jeremy Tibble, Valerie Voon, Hugo D. Critchley, Mara Cercignani, and Neil A. Harrison in Biological Psychiatry. Published online February 1 2016 doi:10.1016/j.biopsych.2015.05.015
Abstract
Acute Changes in Striatal Microstructure Predict the Development of Interferon-Alpha Induced Fatigue
Background
Interferon-alpha (IFN-α) is a key mediator of antiviral immune responses used clinically for hepatitis C treatment. Though effective, IFN-α induces marked behavioral changes that, when severe, can appear indistinguishable from major depression. Curiously, fatigue and motivational impairment evolve rapidly, suggesting acute engagement of immune-brain communicatory pathways, yet mood impairments typically emerge later, after weeks of treatment. Whether this reflects prolonged modulation of motivational processes underpinning fatigue or separate neurobiological mechanisms is currently unclear.
Methods
Here, we used quantitative magnetization transfer (qMT) imaging, an advanced microstructural neuroimaging technique sensitive to effects of inflammation, in a prospective study design to measure acute brain changes to IFN-α and relate these to later development of discrete behavioral changes. Twenty-three patients initiating IFN-α treatment for hepatitis C underwent qMT imaging and blood sampling at baseline and 4 hours after their first IFN-α injection. Comprehensive behavioral and psychological assessments were completed at both scanning sessions and at treatment weeks 4, 8, 12, and 24.
Results
IFN-α injection stimulated an acute inflammatory cytokine response and evoked fatigue that peaked between 4 and 12 weeks, preceding mood change by 4 weeks. In the brain, IFN-α induced an acute change in striatal microstructure that additionally predicted development of fatigue but not mood symptoms.
Conclusions
Our findings highlight qMT as an in vivo biomarker of central effects of peripheral inflammation. We demonstrate exquisite sensitivity of the striatum to IFN-α, implicate striatal perturbation in IFN-α-induced fatigue, and dissociate this from mechanisms underlying IFN-α-induced mood symptoms, providing empirical support for distinct neural substrates mediating actions on motivation and mood.
“Acute Changes in Striatal Microstructure Predict the Development of Interferon-Alpha Induced Fatigue” by Nicholas G. Dowell, Ella A. Cooper, Jeremy Tibble, Valerie Voon, Hugo D. Critchley, Mara Cercignani, and Neil A. Harrison in Biological Psychiatry. Published online February 1 2016 doi:10.1016/j.biopsych.2015.05.015






