Summary: Stress such as intraocular pressure elevation in the eye causes the retinal tissue to undergo transcriptional and epigenetic changes similar to natural aging.
Source: UC Irvine
New research from the University of California, Irvine, suggests aging is an important component of retinal ganglion cell death in glaucoma, and that novel pathways can be targeted when designing new treatments for glaucoma patients.
The study was published today in Aging Cell. Along with her colleagues, Dorota Skowronska‐Krawczyk, Ph.D., assistant professor in the Departments of Physiology & Biophysics and Ophthalmology and the faculty of the Center for Translational Vision Research at the UCI School of Medicine, describes the transcriptional and epigenetic changes happening in aging retina.
The team shows how stress, such as intraocular pressure (IOP) elevation in the eye, causes retinal tissue to undergo epigenetic and transcriptional changes similar to natural aging. And, how in young retinal tissue, repetitive stress induces features of accelerated aging including the accelerated epigenetic age.
Aging is a universal process that affects all cells in an organism. In the eye, it is a major risk factor for a group of neuropathies called glaucoma. Because of the increase in aging populations worldwide, current estimates show that the number of people with glaucoma (aged 40-80) will increase to over 110 million in 2040.
“Our work emphasizes the importance of early diagnosis and prevention as well as age-specific management of age-related diseases, including glaucoma,” said Skowronska-Krawczyk.
“The epigenetic changes we observed suggest that changes on the chromatin level are acquired in an accumulative way, following several instances of stress. This provides us with a window of opportunity for the prevention of vision loss, if and when the disease is recognized early.”
In humans, IOP has a circadian rhythm. In healthy individuals, it oscillates typically in the 12-21 mmHg range and tends to be highest in approximately two thirds of individuals during the nocturnal period.
Due to IOP fluctuations, a single IOP measurement is often insufficient to characterize the real pathology and risk of disease progression in glaucoma patients.
Long-term IOP fluctuation has been reported to be a strong predictor for glaucoma progression. This new study suggests that the cumulative impact of the fluctuations of IOP is directly responsible for the aging of the tissue.
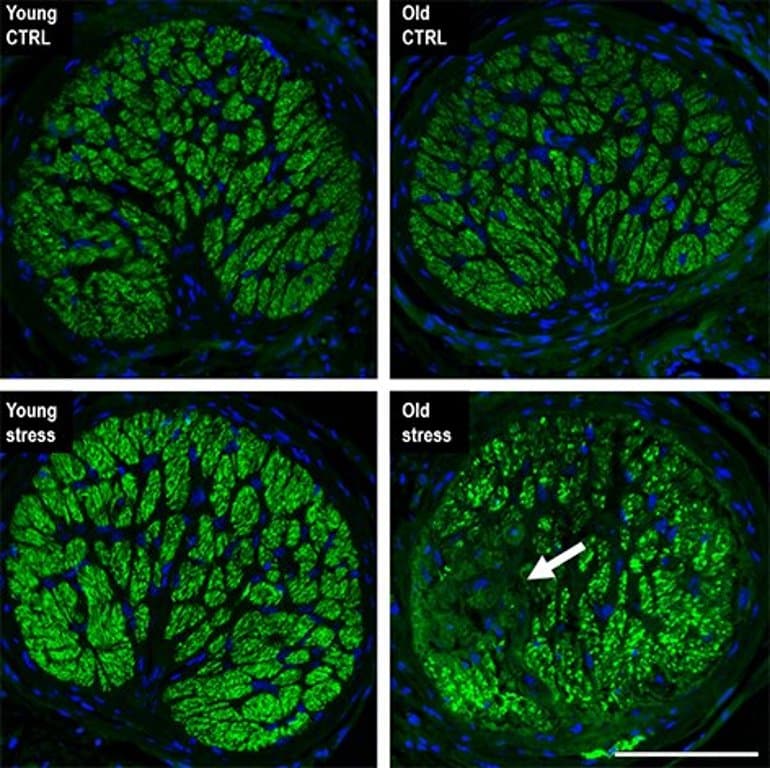
“Our work shows that even moderate hydrostatic IOP elevation results in retinal ganglion cell loss and corresponding visual defects when performed on aged animals,” said Skowronska-Krawczyk.
“We are continuing to work to understand the mechanism of accumulative changes in aging in order to find potential targets for therapeutics. We are also testing different approaches to prevent the accelerated aging process resulting from stress.”
Researchers now have a new tool to estimate the impact of stress and treatment on the aging status of retinal tissue, which has made these new discoveries possible. In collaboration with the Clock Foundation and Steve Horvath, Ph.D., from Altos Labs, who pioneered the development of epigenetic clocks that can measure age based on methylation changes in the DNA of tissues, it was possible for researchers to show that repetitive, mild IOP elevation can accelerate epigenetic age of the tissues.
“In addition to measuring vision decline and some structural changes due to stress and potential treatment, we can now measure the epigenetic age of retinal tissue and use it to find the optimal strategy to prevent vision loss in aging,” said Skowronska-Krawczyk.
About this visual neuroscience research news
Author: Press Office
Source: UC Irvine
Contact: Press Office – UC Irvine
Image: The image is credited to UC Irvine
Original Research: Open access.
“Stress induced aging in mouse eye” by Qianlan Xu et al. Aging Cell
Abstract
Stress induced aging in mouse eye
Aging, a universal process that affects all cells in an organism, is a major risk factor for a group of neuropathies called glaucoma, where elevated intraocular pressure is one of the known stresses affecting the tissue.
Our understanding of molecular impact of aging on response to stress in retina is very limited; therefore, we developed a new mouse model to approach this question experimentally.
Here we show that susceptibility to response to stress increases with age and is primed on chromatin level.
We demonstrate that ocular hypertension activates a stress response that is similar to natural aging and involves activation of inflammation and senescence.
We show that multiple instances of pressure elevation cause aging of young retina as measured on transcriptional and DNA methylation level and are accompanied by local histone modification changes.
Our data show that repeated stress accelerates appearance of aging features in tissues and suggest chromatin modifications as the key molecular components of aging.
Lastly, our work further emphasizes the importance of early diagnosis and prevention as well as age-specific management of age-related diseases, including glaucoma.






