Summary: A new study may help answer the age old questions as to whether animals’ sense of smell became so viable. The findings shed light on why some animals developed extraordinary olfactory systems.
Source: Washington University School of Medicine.
Dogs, known for their extraordinarily keen senses of smell, can be trained to use their sensitive sniffers to find drugs, bombs, bed bugs, missing hikers and even cancer. Among dogs and other animals that rely on smell, at least one factor that may give them an advantage is a sheet of tissue in the nasal cavity.
In humans, this tissue — called the olfactory epithelium — is a single flat sheet lining the roof of the nasal cavity. In dogs, however, the olfactory epithelium forms a complex maze, folding and curling over a number of bony protrusions, called turbinates, that form in the nasal cavity. The olfactory epithelium contains specialized neurons that bind to odor molecules and send signals to the brain that are interpreted as smell. Dogs have hundreds of millions more of these neurons than people do. It is assumed this added structural complexity is responsible for dogs’ superior ability to smell. But, surprisingly, that has never been shown scientifically.
Now, researchers at Washington University School of Medicine in St. Louis have uncovered new details in how the olfactory epithelium develops. The new knowledge could help scientists prove that turbinates and the resulting larger surface area of the olfactory epithelium are one definitive reason dogs smell so well.
“We think the surface area of the sheet matters in how well animals smell and in the types of smells they can detect,” said David M. Ornitz, MD, PhD, the Alumni Endowed Professor of Developmental Biology. “One reason we think this stems from differences in the complexity of these turbinates. Animals that we think of as having a great sense of smell have really complex turbinate systems.”
The study, published Aug. 9 in the journal Developmental Cell, also could help answer a longstanding evolutionary question: How did animals’ senses of smell become so enormously variable? The way these abilities came to diverge over evolutionary history remains a mystery. Understanding these signals could help scientists tease out how dogs evolved an extraordinary olfactory system and humans wound up with a comparatively stunted one.
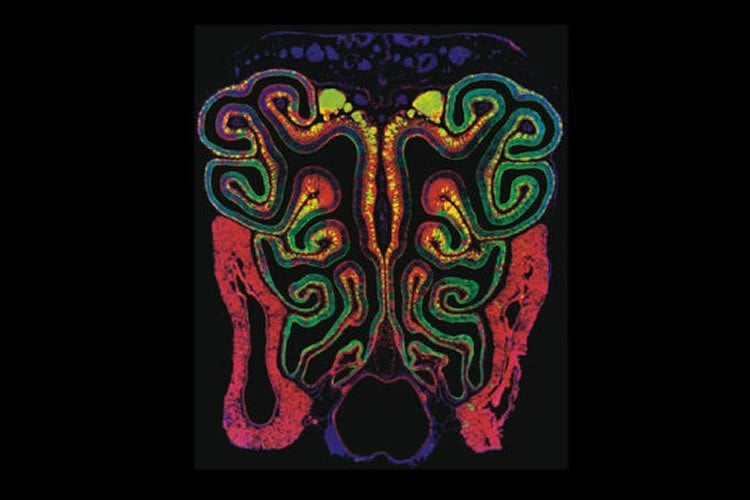
First author Lu M. Yang, a graduate student in Ornitz’s lab, found that a newly discovered stem cell the researchers dubbed FEP cells control the size of the surface area of the olfactory epithelium. These stem cells also send a specific signaling molecule to the underlying turbinates, telling them to grow. The evidence suggests that this signaling crosstalk between the epithelium and the turbinates regulates the scale of the olfactory system that ends up developing, sometimes resulting in olfactory epithelia with larger surface areas, such as in dogs.
When the stem cells can’t signal properly, turbinate growth and olfactory epithelium surface area experience an arrested development. To study this in the lab, mice with such olfactory stunting could, in theory, be compared with typical mice to learn more about how these signals govern the final complexity of an animal’s olfactory system.
“Before our study, we didn’t know how the epithelium expands from a tiny patch of cells to a large sheet that develops in conjunction with complex turbinates,” Yang said. “We can use this to help understand why dogs, for example, have such a good sense of smell. They have extremely complex turbinate structures, and now we know some of the details about how those structures develop.”
Funding: This work was funded by the March of Dimes Foundation; the National Institutes of Health (NIH), grant numbers HL111190 and DC012825; the Department of Developmental Biology at Washington University; the HOPE Center Alafi Neuroimaging Laboratory, grant number NCRR 1S10RR027552; and the Washington University Center for Cellular Imaging of the Children’s Discovery Institute, grant numbers CDI-CORE-2015-505 and NS086741.
Source: Washington University School of Medicine
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Lu Yang.
Original Research: Abstract for “FGF20-Expressing, Wnt-Responsive Olfactory Epithelial Progenitors Regulate Underlying Turbinate Growth to Optimize Surface Area” by Lu M. Yang, Sung-Ho Huh, and David M. Ornitz in Developmental Cell. Published August 9 2018.
doi:10.1016/j.devcel.2018.07.010
[cbtabs][cbtab title=”MLA”]Washington University School of Medicine”How the Sense of Smell Developed.” NeuroscienceNews. NeuroscienceNews, 10 August 2018.
<https://neurosciencenews.com/smell-evolution-9688/>.[/cbtab][cbtab title=”APA”]Washington University School of Medicine(2018, August 10). How the Sense of Smell Developed. NeuroscienceNews. Retrieved August 10, 2018 from https://neurosciencenews.com/smell-evolution-9688/[/cbtab][cbtab title=”Chicago”]Washington University School of Medicine”How the Sense of Smell Developed.” https://neurosciencenews.com/smell-evolution-9688/ (accessed August 10, 2018).[/cbtab][/cbtabs]
Abstract
FGF20-Expressing, Wnt-Responsive Olfactory Epithelial Progenitors Regulate Underlying Turbinate Growth to Optimize Surface Area
The olfactory epithelium (OE) is a neurosensory organ required for the sense of smell. Turbinates, bony projections from the nasal cavity wall, increase the surface area within the nasal cavity lined by the OE. Here, we use engineered fibroblast growth factor 20 (Fgf20) knockin alleles to identify a population of OE progenitor cells that expand horizontally during development to populate all lineages of the mature OE. We show that these Fgf20-positive epithelium-spanning progenitor (FEP) cells are responsive to Wnt/β-Catenin signaling. Wnt signaling suppresses FEP cell differentiation into OE basal progenitors and their progeny and positively regulates Fgf20 expression. We further show that FGF20 signals to the underlying mesenchyme to regulate the growth of turbinates. These studies thus identify a population of OE progenitor cells that function to scale OE surface area with the underlying turbinates.






