Summary: Sleep twitches help enrich the encoding of sensory information in newborn rats, laying the groundwork for later motor function.
Source: SfN
Electrical activity in the motor cortex of rats transforms from redundant to complex over the span of four days shortly after birth. Sleep twitches guide this metamorphosis, according to new research published in Journal of Neuroscience.
Despite its name, the motor cortex doesn’t control movement right off the bat. Early in development, this part of the brain is solely a sensory structure.
Feedback from self-generated movements — sleep twitches in particular — may build representations of the body that will later orchestrate movement.
To characterize this transition, Glanz et al. recorded electrical activity from the motor cortex of rat pups eight and 12 days after birth while monitoring their behavior. On both days, sleep twitches drove more electrical activity than wake movements.
Twitches on postnatal day eight (P8) activated many neurons at the same time — there was high redundance. This redundance disappeared by P12: each twitch activated a smaller group of neurons, a sign of maturing circuits.
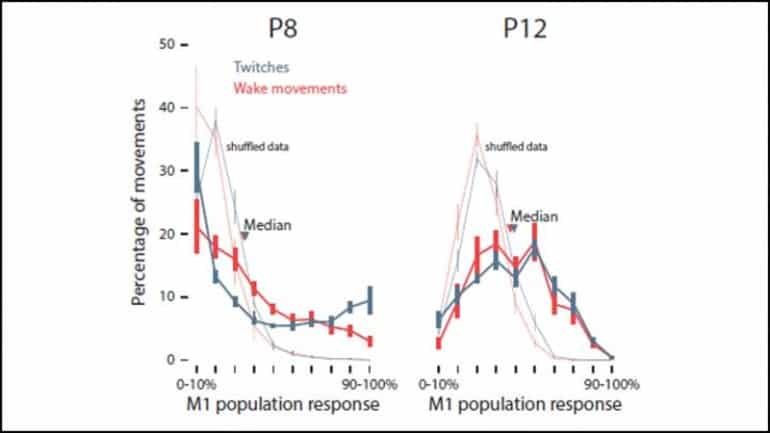
Larger twitches induced larger responses on P8, a trend not seen on P12. Yet the motor cortex became more sensitive to small twitches by P12.
These changes mark a key developmental transition in the motor cortex facilitated by sleep twitches.
About this neuroscience research news
Source: SfN
Contact: Calli McMurray – SfN
Image: The image is credited to Glanz et al., JNeurosci 2021
Original Research: Closed access.
“Sensory coding of limb kinematics in motor cortex across a key developmental transition” by Ryan M. Glanz, James C. Dooley, Greta Sokoloff and Mark S. Blumberg. Journal of Neuroscience
Abstract
Sensory coding of limb kinematics in motor cortex across a key developmental transition
Primary motor cortex (M1) undergoes protracted development in mammals, functioning initially as a sensory structure. Throughout the first postnatal week in rats, M1 is strongly activated by self-generated forelimb movements—especially by the twitches that occur during active sleep.
Here, we quantify the kinematic features of forelimb movements to reveal receptive-field properties of individual units within the forelimb region of M1. At postnatal day (P) 8, nearly all units were strongly modulated by movement amplitude, especially during active sleep. By P12, only a minority of units continued to exhibit amplitude-tuning, regardless of behavioral state. At both ages, movement direction also modulated M1 activity, though to a lesser extent.
Finally, at P12, M1 population-level activity became more sparse and decorrelated, along with a substantial alteration in the statistical distribution of M1 responses to limb movements.
These findings reveal a transition toward a more complex and informationally rich representation of movement long before M1 develops its motor functionality.
SIGNIFICANCE STATEMENT:
Primary motor cortex (M1) plays a fundamental role in the generation of voluntary movements and motor learning in adults. In early development, however, M1 functions as a prototypical sensory structure.
Here, we demonstrate in infant rats that M1 codes for the kinematics of self-generated limb movements long before M1 develops its capacity to drive movements themselves. Moreover, we identify a key transition during the second postnatal week in which M1 activity becomes more informationally complex.
Together, these findings further delineate the complex developmental path by which M1 develops its sensory functions in support of its later-emerging motor capacities.






