Summary: McGill researchers report RNA that encodes synaptic proteins degrades more rapidly in Alzheimer’s patients.
Source: McGill University.
McGill University researchers have discovered a cellular mechanism that may contribute to the breakdown of communication between neurons in Alzheimer’s disease.
Their study, published in Nature Communications, homes in on the role of RNA molecules involved in synaptic transmission –- the process through which neurons communicate with each other. In the brain tissue of Alzheimer’s patients, the RNAs that encode synaptic proteins are degraded more rapidly than in healthy brain cells, the researchers found. They also discovered that a protein that helps stabilize these RNAs was less abundant in the neurons of Alzheimer’s patients.
Taken together, these findings indicate that inadequate levels of the protein, known as RBFOX1, may be a factor in the faulty connections that are a hallmark of Alzheimer’s disease, says Hamed S. Najafabadi, senior author of the paper and an assistant professor in McGill’s Department of Human Genetics.
A new piece of the puzzle
Although Alzheimer’s is by far the most common form of dementia, its underlying mechanisms aren’t fully understood, and there are currently no treatments to stop its progression. The McGill study reveals a new piece of the puzzle, which could provide leads for new therapeutic approaches.
Human cells produce thousands of different types of RNAs to carry genetic information. RNAs also decay constantly, and the balance between production and degradation determines how much of a given RNA is present in the cell. Yet scientists know relatively little about how RNA decay is controlled, largely because methods of measuring degradation have been expensive and not applicable to human tissues.
Previous research by Najafabadi had shown that RNA degradation is involved in different human diseases. Most of those findings came from studies in cell line models of diseases, however. “We wanted to directly measure the RNA degradation rate in human tissues, but available methods weren’t able to do that,” Najafabadi explains. So his team hit on a way to get around that problem. “We realized that by modeling the process of RNA production and decay, we could devise a mathematical method to calculate RNA degradation using existing genomics technologies.”
Measuring RNA decay
To test their new approach, the McGill researchers enlisted the help of scientists at the University of California, San Francisco. The California team, led by Hani Goodarzi, grew cells in a lab, and measured the rate of RNA degradation using a conventional method. At the same time, the McGill researchers estimated the rate using their mathematical method. The two results matched up, validating the mathematical framework.
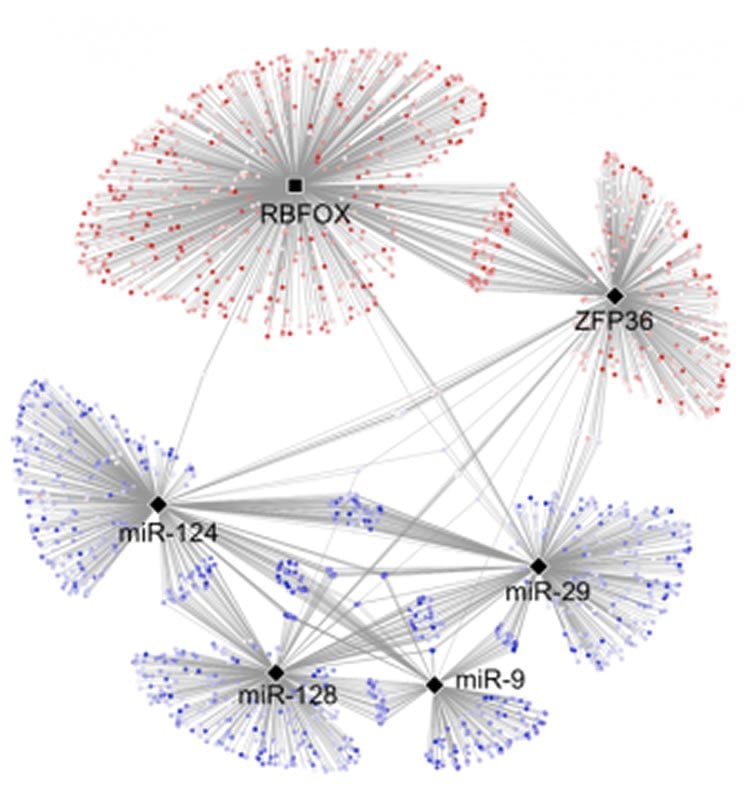
Najafabadi and McGill graduate student Rached Alkallas then applied the mathematical method to analyze publicly available data on the brain tissues of people who died from Alzheimer’s. They also analyzed brain tissues from people who didn’t suffer from Alzheimer’s. Comparing those two groups revealed the rapid rate of RNA degradation and short supply of RBFOX1 protein in the Alzheimer’s patients.
“There’s still a lot to learn about the role of RNA degradation in Alzheimer’s and other diseases,” Najafabadi says. “For example, why is there a reduction of RBFOX1 in Alzheimer’s? Is the reduced amount of this protein a risk factor, or a feature of the later stages of the disease? And can we restore at least part of the normal function of the neurons by controlling the activity of RBFOX1?”
Funding: Funding for this work was provided by the McGill University Faculty of Medicine and the National Cancer Institute (U.S.).
Source: Chris Chipello – McGill University
Image Source: NeuroscienceNews.com images iscredited to the researchers.
Original Research: Full open access research for “Inference of RNA decay rate from transcriptional profiling highlights the regulatory programs of Alzheimer’s disease” by Rached Alkallas, Lisa Fish, Hani Goodarzi & Hamed S. Najafabadi in Nature Communications. Published online October 13 2017 doi:10.1038/s41467-017-00867-z
[cbtabs][cbtab title=”MLA”]McGill University “New Mechanism Detected in Alzheimer’s Disease.” NeuroscienceNews. NeuroscienceNews, 13 October 2017.
<https://neurosciencenews.com/rna-alzheimers-genetics-7739/>.[/cbtab][cbtab title=”APA”]McGill University (2017, October 13). New Mechanism Detected in Alzheimer’s Disease. NeuroscienceNews. Retrieved October 13, 2017 from https://neurosciencenews.com/rna-alzheimers-genetics-7739/[/cbtab][cbtab title=”Chicago”]McGill University “New Mechanism Detected in Alzheimer’s Disease.” https://neurosciencenews.com/rna-alzheimers-genetics-7739/ (accessed October 13, 2017).[/cbtab][/cbtabs]
Abstract
Inference of RNA decay rate from transcriptional profiling highlights the regulatory programs of Alzheimer’s disease
The abundance of mRNA is mainly determined by the rates of RNA transcription and decay. Here, we present a method for unbiased estimation of differential mRNA decay rate from RNA-sequencing data by modeling the kinetics of mRNA metabolism. We show that in all primary human tissues tested, and particularly in the central nervous system, many pathways are regulated at the mRNA stability level. We present a parsimonious regulatory model consisting of two RNA-binding proteins and four microRNAs that modulate the mRNA stability landscape of the brain, which suggests a new link between RBFOX proteins and Alzheimer’s disease. We show that downregulation of RBFOX1 leads to destabilization of mRNAs encoding for synaptic transmission proteins, which may contribute to the loss of synaptic function in Alzheimer’s disease. RBFOX1 downregulation is more likely to occur in older and female individuals, consistent with the association of Alzheimer’s disease with age and gender.
“Inference of RNA decay rate from transcriptional profiling highlights the regulatory programs of Alzheimer’s disease” by Rached Alkallas, Lisa Fish, Hani Goodarzi & Hamed S. Najafabadi in Nature Communications. Published online October 13 2017 doi:10.1038/s41467-017-00867-z