Summary: Researchers uncover how the SNO reaction may contribute to the onset of Parkinson’s disease.
Source: Scripps Research Institute.
A new study from The Scripps Research Institute (TSRI) is the first to show precisely how a process in nerve cells called the S-nitrosylation (SNO) reaction–which can be caused by aging, pesticides and pollution–may contribute to Parkinson’s disease.
The leader of the study, TSRI Professor Stuart Lipton, M.D., Ph.D., is now investigating how new pharmaceuticals might counteract this harmful S-nitrosylation reaction.
“The new finding gives us a clue as to where to intervene,” said Lipton, who holds a joint position at the University of California, San Diego, School of Medicine, where he is a practicing clinical neurologist involved in the care of Parkinson’s patients.
The study was published today in the journal Cell Reports.
‘PINK-SNO Man’ Implicated in Parkinson’s
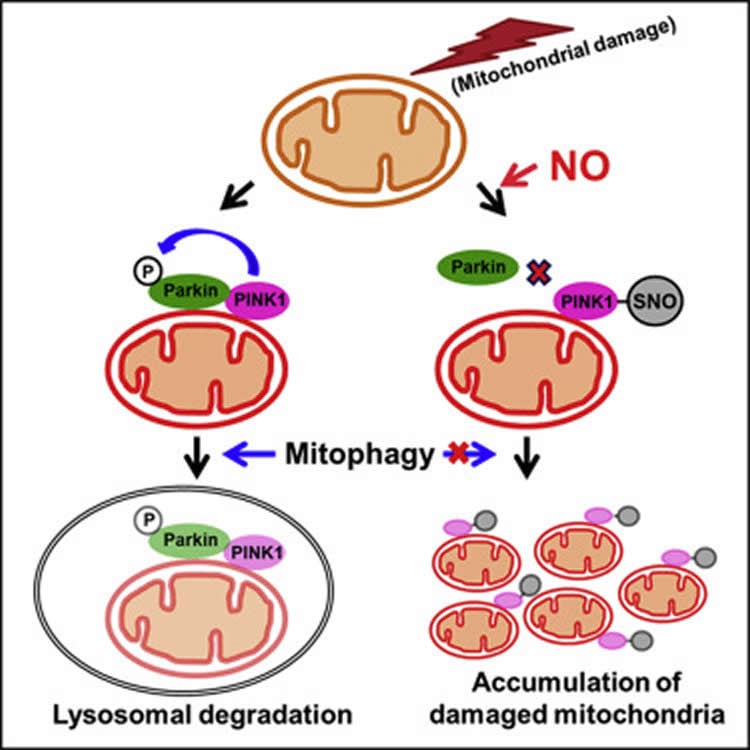
The SNO reaction attaches a nitric oxide-like molecule to protein called PINK1, where the molecule attaches to a building block on PINK1 called a cysteine residue. This changes PINK1’s activity–and its ability to do its job.
Since Lipton’s group co-discovered the SNO reaction some 20 years ago, scientists have linked the reaction to protein misfolding and nerve cell damage in cases of Alzheimer’s, Huntington’s, amyotrophic lateral sclerosis (ALS/Lou Gehrig’s disease) and Parkinson’s disease, as well as heart/cardiovascular disease and cancer.
In the new study, Lipton and his colleagues used human stem cell and mouse models to show exactly how SNO can trigger cell death in Parkinson’s disease. They found that when SNO modifies PINK1, nerve cells cannot recruit another protein called Parkin to get rid of damaged mitochondria.
“Mitochondria are the energy powerhouses of the cell,” explained Lipton. Because neurons need a lot of energy, Lipton said, it is especially crucial for them to use only healthy mitochondria and get rid of the damaged ones. Mitochondria can be damaged as people age and cells experience various forms of stress, and it is the job of PINK1 to help trigger a process called mitophagy to remove those dysfunctional mitochondria.
Previous studies had shown that inherited mutations to the gene that codes for PINK1 can stop a person from making working versions of the protein. This means their neurons cannot clear damaged mitochondria, and those cells eventually die–which can cause Parkinson’s.
The SNO reaction seems to cause this same problem, but it is not inherited. Instead, cells start “SNO-ing” proteins when they get overwhelmed by reactive nitrogen molecules. “The quantities of these reactive chemical species get so high that cells start SNO-ing proteins, like PINK1, that would normally not be SNO-ed,” said Lipton. The researchers call this the PINK-SNO complex, or a “PINK-SNO man.”
“Formation of PINK-SNO is definitely harmful to nerve cells in the Parkinson’s brain,” said Lipton.
So where are these reactive nitrogen species coming from? The scientists noted that cells can generate excessive nitric oxide in response to pesticide exposure, other toxins, and possibly even air pollution.
“This is a scary thought, but also a hopeful thought,” said Lipton. “if we can figure out how we’re doing this to ourselves, we may be able to control it.”
The new study adds to the evidence that some degenerative brain diseases appear to be caused by a combination of genetics and environment. Lipton explained that genetics may leave some people “predisposed” to be at risk for SNO-related Parkinson’s.
Because humans inherit two copies (one from each parent) of the gene that encodes PINK1, we all have at least one copy of the gene if the other is mutated. Depending on the protein, this may or may not be sufficient for normal function.
“But, in any event, if the protein translated from the remaining good copy of the gene is then targeted by SNO, then you are stuck making dysfunctional PINK1 even from the remaining good copy of the gene,” said Lipton. “The take-home message here is that the environment may affect you based on your individual genetics, and thus both are influential in causing diseases like Parkinson’s.”
Interestingly, Lipton’s team found that SNO-ing appears to occur early in disease progression–early enough that intervention may be able to save brain function. He said the next step is to study how we can prevent these aberrant SNO reactions on particular proteins like PINK1.
In addition to Lipton, other authors of the study, “S-Nitrosylation of PINK1 Attenuates PINK1/Parkin-Dependent Mitophagy in hiPSC-Based Parkinson’s Disease Models,” include first author Chang-Ki Oh, Nima Dolatabadi, Daniel B. McClatchy, Jolene K. Diedrich, John R. Yates, and Tomohiro Nakamura of TSRI; Abdullah Sultan and Rajesh Ambasudhan of the Scintillon Institute in San Diego; and Frank Soldner and Rudolf Jaenisch of the Massachusetts Institute of Technology/Whitehead Institute.
Funding: This research was supported by the National Institutes of Health (grants R01 NS086890, P01 ES016738, DP1 DA041722, RF1 AG057409, R01 AG056259, P30 NS076411, P41 GM103533, R37 HD045022, R37 CA084198 and R01 NS088538), a Distinguished Investigator Award from the Brain & Behavior Research Foundation, and the Michael J. Fox Foundation.
Source: Madeline McCurry-Schmidt – Scripps Research Institute
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to the researchers/Cell Reports.
Original Research: Full open access research for “S-Nitrosylation of PINK1 Attenuates PINK1/Parkin-Dependent Mitophagy in hiPSC-Based Parkinson’s Disease Models” by Chang-Ki Oh, Abdullah Sultan, Joseph Platzer, Nima Dolatabadi, Frank Soldner, Daniel B. McClatchy, Jolene K. Diedrich, John R. Yates III, Rajesh Ambasudhan, Tomohiro Nakamura, Rudolf Jaenisch, and Stuart A. Lipton in Cell Reports. Published online November 21 2017 doi:10.1016/j.celrep.2017.10.068
[cbtabs][cbtab title=”MLA”]Scripps Research Institute “Good Cells Gone Bad.” NeuroscienceNews. NeuroscienceNews, 21 November 2017.
<https://neurosciencenews.com/pink-sno-parkinsons-7998/>.[/cbtab][cbtab title=”APA”]Scripps Research Institute (2017, November 21). Good Cells Gone Bad. NeuroscienceNews. Retrieved November 21, 2017 from https://neurosciencenews.com/pink-sno-parkinsons-7998/[/cbtab][cbtab title=”Chicago”]Scripps Research Institute “Good Cells Gone Bad.” https://neurosciencenews.com/pink-sno-parkinsons-7998/ (accessed November 21, 2017).[/cbtab][/cbtabs]
Abstract
S-Nitrosylation of PINK1 Attenuates PINK1/Parkin-Dependent Mitophagy in hiPSC-Based Parkinson’s Disease Models
Highlights
•Nitric oxide inhibits PINK1 kinase activity via S-nitrosylation of PINK1 at Cys568
•S-Nitrosylation of PINK1 decreases translocation of Parkin to mitochondria
•S-Nitrosylation of PINK1 thus impairs mitophagy in hiPSC-derived neurons
•S-Nitrosylation of PINK1 exacerbates neuronal death in Parkinson’s disease models
Summary
Mutations in PARK6 (PINK1) and PARK2 (Parkin) are linked to rare familial cases of Parkinson’s disease (PD). Mutations in these genes result in pathological dysregulation of mitophagy, contributing to neurodegeneration. Here, we report that environmental factors causing a specific posttranslational modification on PINK1 can mimic these genetic mutations. We describe a molecular mechanism for impairment of mitophagy via formation of S-nitrosylated PINK1 (SNO-PINK1). Mitochondrial insults simulating age- or environmental-related stress lead to increased SNO-PINK1, inhibiting its kinase activity. SNO-PINK1 decreases Parkin translocation to mitochondrial membranes, disrupting mitophagy in cell lines and human-iPSC-derived neurons. We find levels of SNO-PINK1 in brains of α-synuclein transgenic PD mice similar to those in cell-based models, indicating the pathophysiological relevance of our findings. Importantly, SNO-PINK1-mediated deficits in mitophagy contribute to neuronal cell death. These results reveal a direct molecular link between nitrosative stress, SNO-PINK1 formation, and mitophagic dysfunction that contributes to the pathogenesis of PD.
“S-Nitrosylation of PINK1 Attenuates PINK1/Parkin-Dependent Mitophagy in hiPSC-Based Parkinson’s Disease Models” by Chang-Ki Oh, Abdullah Sultan, Joseph Platzer, Nima Dolatabadi, Frank Soldner, Daniel B. McClatchy, Jolene K. Diedrich, John R. Yates III, Rajesh Ambasudhan, Tomohiro Nakamura, Rudolf Jaenisch, and Stuart A. Lipton in Cell Reports. Published online November 21 2017 doi:10.1016/j.celrep.2017.10.068






