Summary: Combining niacin, or vitamin B3, with chemotherapy, helped slow the progression and increase life span in mouse models of glioblastoma.
Source: University of Calgary
A new study by members of the Cumming School of Medicine (CSM) at the University of Calgary finds niacin, commonly called vitamin B3, combined with chemotherapy can help immune cells attack glioblastoma (a type of brain tumour), dramatically slowing progression of the disease, in mice. The results published in Science Translational Medicine found the lifespan of mice with glioblastoma that received combination therapy tripled, increasing to 150 days from 40 days.
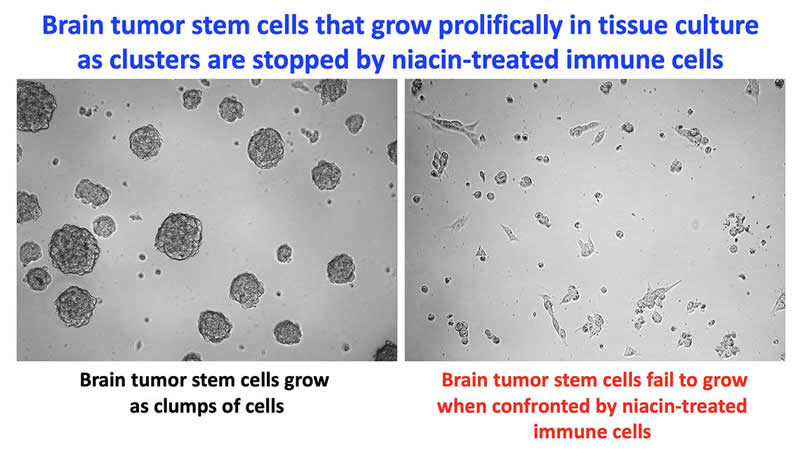
“It is a remarkable result. While it’s not a cure, it’s a promising step forward against this incurable disease,” says Dr. Wee Yong, PhD, the principal investigator on the study and a professor in the departments of Clinical Neurosciences and Oncology at the CSM and member of the Hotchkiss Brain Institute and Arnie Charbonneau Cancer Institute. “The brain tumour stem cells for glioblastoma have been very resistant to treatment, so instead of targeting those cells we targeted the immune system to help the body to attack and destroy the stem cells.”
Glioblastoma is the most aggressive form of brain cancer. Even with treatment, chemotherapy and radiation, most people die within 14 to 16 months of being diagnosed. One of the reasons this cancer is so deadly is because it hijacks the immune system, suppressing it and reprogramming immune cells to work for the tumour.
In the study, the researchers found that niacin therapy alone extended survival and that the combination therapy with temozolomide (a chemotherapy drug commonly used against glioblastoma) markedly prolonged survival by stimulating and re-educating immune cells to stop helping the cancer and instead, destroy it.
“We were able to help immune cells do what they’re supposed to do, attack and kill cancer cells,” says Dr. Susobhan Sarkar, PhD, first author on the study. “We screened 1,040 compounds and found niacin had the properties needed to activate immune cells, specifically myeloid cells, and inhibit the growth of brain tumour initiating stem cells.”
The study is supported by Alberta Innovates in collaboration with the Alberta Cancer Foundation and the HBI through a translational research grant donated by the Ronald and Irene Ward Foundation and the Canadian Institutes of Health Research (CIHR). The CIHR has already provided funding to move this research forward to a clinical trial.
“We are very fortunate to have the support of the CIHR,” says Yong. “We still require approvals from Health Canada and ethics. It’s extremely important to follow strict protocols and conduct a clinical trial first, even though this treatment involves two well-known, existing therapies. It’s important people don’t rush out and try adding niacin on their own, as we need to confirm dosage, delivery and length of time for optimum clinical results.”
Yong will be working with Dr. Gloria Roldan Urgoiti, MD, an oncologist and Dr. Paula de Robles, MD, a neuro-oncologist at Tom Baker Cancer Centre, on the clinical trial.
Source:
University of Calgary
Media Contacts:
Kelly Johnston – University of Calgary
Image Source:
The image is credited to Wee Yong.
Original Research: Closed access
“”Control of brain tumor growth by reactivating myeloid cells with niacin”. Wee Yong et al.
Science Translational Medicine doi:10.1126/scitranslmed.aay9924.
Abstract
Control of brain tumor growth by reactivating myeloid cells with niacin
Glioblastomas are generally incurable partly because monocytes, macrophages, and microglia in afflicted patients do not function in an antitumor capacity. Medications that reactivate these macrophages/microglia, as well as circulating monocytes that become macrophages, could thus be useful to treat glioblastoma. We have discovered that niacin (vitamin B3) is a potential stimulator of these inefficient myeloid cells. Niacin-exposed monocytes attenuated the growth of brain tumor–initiating cells (BTICs) derived from glioblastoma patients by producing anti-proliferative interferon-α14. Niacin treatment of mice bearing intracranial BTICs increased macrophage/microglia representation within the tumor, reduced tumor size, and prolonged survival. These therapeutic outcomes were negated in mice depleted of circulating monocytes or harboring interferon-α receptor–deleted BTICs. Combination treatment with temozolomide enhanced niacin-promoted survival. Monocytes from glioblastoma patients had increased interferon-α14 upon niacin exposure and were reactivated to reduce BTIC growth in culture. We highlight niacin, a common vitamin that can be quickly translated into clinical application, as an immune stimulator against glioblastomas.






