Simple animal models provide new insight into basic brain function.
Amita Sehgal, PhD, a professor of Neuroscience at the Perelman School of Medicine, University of Pennsylvania, describes in Cell a circuit in the brain of fruit flies that controls their daily, rhythmic behavior of rest and activity. The new study also found that the fly version of the human brain protein known as corticotrophin releasing factor (CRF) is a major coordinating molecule in this circuit. Fly CRF, called DH44, is required for rest/activity cycles and is produced in cells that receive input from the clock cells in the fly brain. In mammals, CRF is secreted rhythmically and it drives the expression of glucocorticoids such as cortisol and is associated with stress and anxiety.
Animal models like flies are helping to fill gaps in current knowledge about how the brain works, notes Sehgal. Indeed, she says, the Brain Research through Advancing Innovative Neurotechnologies (BRAIN), initiative, a project of the National Institutes of Health, includes the study of simple animal models, which are expected to provide more detailed insight into brain function.
Though much is known about the cellular and molecular components of the clock, the connections that link clock cells to overt behaviors, such as rest/activity behavior, have not been identified. “This study is essentially a map-of-the-circuitry experiment,” says Sehgal, who is also an investigator with the Howard Hughes Medical Institute (HHMI). Like humans, flies are active during the day — walking, flying, feeding and mating — and spend most of the night asleep.
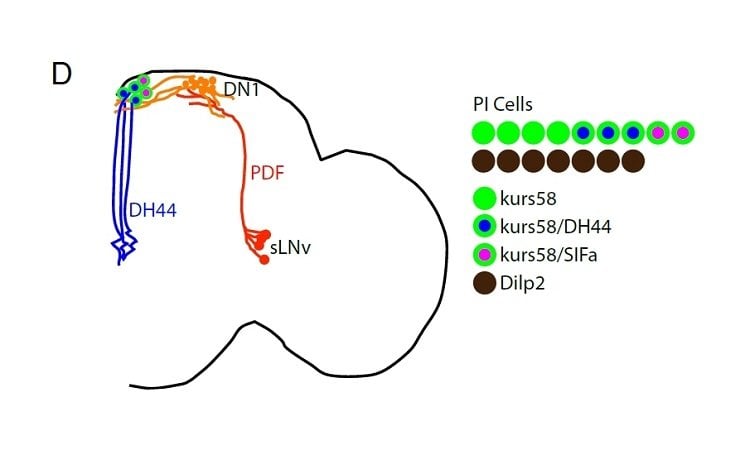
“We conducted a screen for circadian-relevant neurons in the flybrain and found that cells of the pars intercerebralis — the fly version of the mammalian hypothalamus — comprise an important component of the circadian output pathway for rest/activity rhythms in flies,” Sehgal says. The mammalian hypothalamus is a neuroendocrine structure that regulates sleep, circadian rhythms, feeding and, metabolism.
The Penn team did a random targeting of cells, activating neuronal firing with a transgene designed for this purpose, to see which cells are important in the rest/active behavior. They found that cells in the pars intercerebralis (PI) are essential for rhythmic behavior, and PI cells are connected to the clock cells through a circuit of at least two synapses.
Molecular profiling of PI cells identified the fly version of DH44 as a circadian molecule that is specifically expressed by PI neurons and required for normal rest/activity rhythms in flies. And, when the scientists selectively activated or removed just six PI cells positive for DH44, the fly’s activity cycles became irregular. In other words, the flies no longer restricted their sleep to the dark and their activity to the light, but instead showed more random distribution of these behaviors.
Coauthors are Daniel J. Cavanaugh, Jill D. Geratowski, Julian R. A. Wooltorton, Jennifer M.
Spaethling, Xiangzhong Zheng, and James H. Eberwine, all from Penn, and Clare E. Hector and Erik C. Johnson, from Wake Forest University.
This research as funded in part by the National Institute of Neurological Disorders and Stroke (2R01NS048471) and HHMI.
Contact: Karen Kreeger – University of Pennsylvania
Source: University of Pennsylvania press release
Image Source: The image is credited to Daniel Cavanaugh and is adapted from the University of Pennsylvania press release
Original Research: Abstract for “Identification of a Circadian Output Circuit for Rest:Activity Rhythms in Drosophila” by Daniel J. Cavanaugh, Jill D. Geratowski, Julian R.A. Wooltorton, Jennifer M. Spaethling, Clare E. Hector, Xiangzhong Zheng, Erik C. Johnson, James H. Eberwine, Amita Sehgal in Cell. Published online April 24 2014 doi:10.1016/j.cell.2014.02.024