Summary: A new study links hippocampal inflammation in multiple sclerosis with an increased risk of developing depression.
Source: Elsevier.
Patients with multiple sclerosis have higher rates of depression than the general population, including people with other life-long disabling diseases. Symptoms of multiple sclerosis arise from an abnormal response of the body’s immune system. Immune response has also been linked to depression, leading researchers to think it could be a shared pathological mechanism that leads to the increased rates of depressive symptoms in patients with multiple sclerosis.
A new study in Biological Psychiatry supports this hypothesis, providing evidence that inflammation of the hippocampus, a region of the brain implicated in the genesis and maintenance of depression and in the pathology of multiple sclerosis, alters its function and contributes to symptoms of depression.
“This study elegantly links hippocampal inflammation to depression,” said Dr. John Krystal, Editor of Biological Psychiatry.
The research was a collaboration between King’s College London, Imperial College London, and Imanova Center for Imaging Sciences. Led by senior authors Paul Matthews and Eugenii Rabiner, the research team combined two complementary brain imaging techniques to study the relationship between hippocampal immune response, functional connections, and depressive symptoms in 13 patients with multiple sclerosis and 22 healthy control subjects. Positron emission tomography (PET) allowed for quantification of activated microglia, a measure of immune response. Functional magnetic resonance imaging (fMRI) assessed the strength of hippocampal connections to an extensive network of brain regions involved in emotion.
First author Dr. Alessandro Colasanti, of King’s College London, explained that PET imaging revealed immune activation in the hippocampus of patients with multiple sclerosis. “We also discovered that more inflammation was associated to more severe symptoms of depression,” said Colasanti.
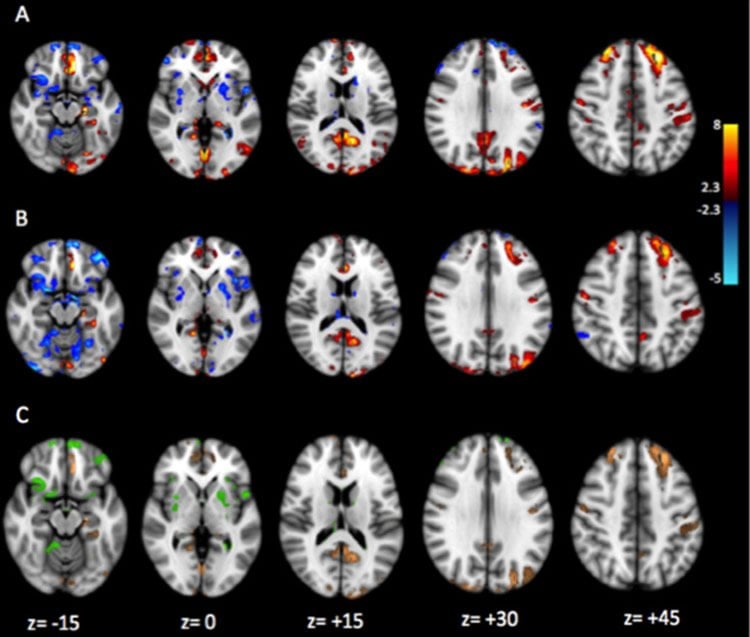
Measurements of functional brain connections with fMRI during rest showed that immune activation in the hippocampus altered its connections with other brain regions. “This study, combining two advanced complementary brain imaging methods, suggests that the inflammation of the hippocampus affects the brain function and causes depression,” said Colasanti.
The findings suggest that hippocampal inflammation could be the contributing cause of high rates of depression in multiple sclerosis. The authors predict that an effective and targeted treatment of brain inflammation would help to restore brain function and protect against depression in multiple sclerosis.
Funding: Open access funded by Wellcome Trust.
The authors’ affiliations, and disclosures of financial and conflicts of interests are available in the article.
John H. Krystal, M.D., is Chairman of the Department of Psychiatry at the Yale University School of Medicine, Chief of Psychiatry at Yale-New Haven Hospital, and a research psychiatrist at the VA Connecticut Healthcare System. His disclosures of financial and conflicts of interests are available here.
Source: Rhiannon Bugno – Elsevier
Image Source: This NeuroscienceNews.com image is credited to Colasanti et al./Biological Psychiatry.
Original Research: Full open access research for “Hippocampal Neuroinflammation, Functional Connectivity, and Depressive Symptoms in Multiple Sclerosis” by Alessandro Colasanti, Qi Guo, Paolo Giannetti, Matthew B. Wall, Rexford D. Newbould, Courtney Bishop, Mayca Onega, Richard Nicholas, Olga Ciccarelli, Paolo A. Muraro, Omar Malik, David R. Owen, Allan H. Young, Roger N. Gunn, Paola Piccini, Paul M. Matthews, and Eugenii A. Rabiner in Biological Psychiatry. Published online April 27 2016 doi:10.1016/j.biopsych.2015.11.022
[cbtabs][cbtab title=”MLA”]Elsevier. “Brain Inflammation Linked to Depression in Multiple Sclerosis.” NeuroscienceNews. NeuroscienceNews, 7 July 2016.
<https://neurosciencenews.com/ms-depression-neuroinflammation-4633/>.[/cbtab][cbtab title=”APA”]Elsevier. (2016, July 7). Brain Inflammation Linked to Depression in Multiple Sclerosis. NeuroscienceNews. Retrieved July 7, 2016 from https://neurosciencenews.com/ms-depression-neuroinflammation-4633/[/cbtab][cbtab title=”Chicago”]Elsevier. “Brain Inflammation Linked to Depression in Multiple Sclerosis.” https://neurosciencenews.com/ms-depression-neuroinflammation-4633/ (accessed July 7, 2016).[/cbtab][/cbtabs]
Abstract
Hippocampal Neuroinflammation, Functional Connectivity, and Depressive Symptoms in Multiple Sclerosis
Background
Depression, a condition commonly comorbid with multiple sclerosis (MS), is associated more generally with elevated inflammatory markers and hippocampal pathology. We hypothesized that neuroinflammation in the hippocampus is responsible for depression associated with MS. We characterized the relationship between depressive symptoms and hippocampal microglial activation in patients with MS using the 18-kDa translocator protein radioligand [18F]PBR111. To evaluate pathophysiologic mechanisms, we explored the relationships between hippocampal neuroinflammation, depressive symptoms, and hippocampal functional connectivities defined by resting-state functional magnetic resonance imaging.
Methods
The Beck Depression Inventory (BDI) was administered to 11 patients with MS and 22 healthy control subjects before scanning with positron emission tomography and functional magnetic resonance imaging. We tested for higher [18F]PBR111 uptake in the hippocampus of patients with MS relative to healthy control subjects and examined the correlations between [18F]PBR111 uptake, BDI scores, and hippocampal functional connectivities in the patients with MS.
Results
Patients with MS had an increased hippocampal [18F]PBR111 distribution volume ratio relative to healthy control subjects (p = .024), and the hippocampal distribution volume ratio was strongly correlated with the BDI score in patients with MS (r = .86, p = .006). Hippocampal functional connectivities to the subgenual cingulate and prefrontal and parietal regions correlated with BDI scores and [18F]PBR111 distribution volume ratio.
Conclusions
Our results provide evidence that hippocampal microglial activation in MS impairs the brain functional connectivities in regions contributing to maintenance of a normal affective state. Our results suggest a rationale for the responsiveness of depression in some patients with MS to effective control of brain neuroinflammation. Our findings also lend support to further investigation of the role of inflammatory processes in the pathogenesis of depression more generally.
“Titi semantics: Context and meaning in Titi monkey call sequences” by Philippe Schlenker, Emmanuel Chemla, Cristiane Cäsar, Robin Ryder, and Klaus Zuberbühler in Natural Language & Linguistic Theory. Published online April 27 2016 doi:10.1007/s11049-016-9337-96t






