Prion diseases–incurable, ultimately fatal, transmissible neurodegenerative disorders of mammals–are believed to develop undetected in the brain over several years from infectious prion protein. In a new study, National Institutes of Health (NIH) scientists report they can detect infectious prion protein in mouse brains within a week of inoculation. Equally surprising, the protein was generated outside blood vessels in a place in the brain where scientists believe drug treatment could be targeted to prevent disease. The study, from NIH’s National Institute of Allergy and Infectious Diseases (NIAID), appears in the Sept. 22 issue of mBio.
Scientists believe prion diseases potentially could be treated if therapy starts early in the disease cycle. However, identifying who needs treatment and pinpointing the optimal timeframe for treatment are open questions for researchers.
Human prion diseases include variant, familial and sporadic Creutzfeldt-Jakob disease (CJD). The most common form, sporadic CJD, affects an estimated one in one million people annually worldwide. Other prion diseases include scrapie in sheep, chronic wasting disease in deer, elk and moose, and bovine spongiform encephalopathy in cattle.
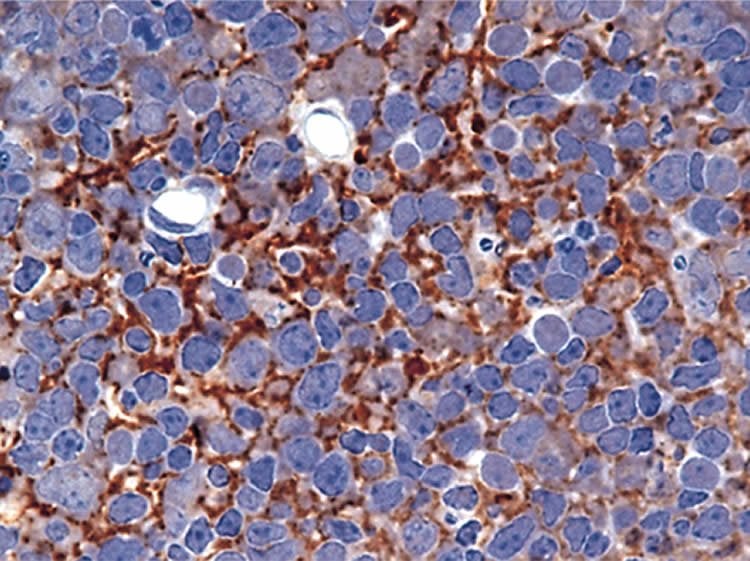
In their study, the NIAID scientists injected infectious scrapie prion protein into the brains of mice. After 30 minutes, they began observing whether the injected material generated new infectious protein at the injection site. By examining mouse brain tissue, the researchers measured and detected new infectious prion protein three days after infection on the outside walls of capillaries and other blood vessels at the injection site. Using Real-Time Quaking-Induced Conversion (RT-QuIC), a feasible testing method for people, the scientists detected newly generated prion protein after seven days. In prior studies, it took about six weeks to detect infectious prion protein. The new findings enhance scientific understanding of where infectious prion diseases might take hold in the brain and provide possible targets for treatment.
Funding: Galina Schmunk, Bryan Boubion and Ian Smith of UCI contributed to the study, which received support from the National Institutes of Health (grants GM048071 and GM1000201) and the William & Nancy Thompson Family Foundation. The discovery is being patented by the University of California as a diagnostic tool and for identifying potential therapeutic agents.
Source: Ken Pekoc – NIH/NIAID
Image Source: The image is credited to McGovern et al./PLOS ONE and is licensed CC BY 2.5
Original Research: Abstract for “Early Generation of New PrPSc on Blood Vessels after Brain Microinjection of Scrapie in Mice” by Bruce Chesebro, James Striebel, Alejandra Rangel, Katie Phillips, Andrew Hughson, Byron Caughey, and Brent Race in mBio. Published online September 22 2015 doi:10.1128/mBio.01419-15
Abstract
Early Generation of New PrPSc on Blood Vessels after Brain Microinjection of Scrapie in Mice
Aggregation of misfolded host proteins in the central nervous system is believed to be important in the pathogenic process in several neurodegenerative diseases of humans, including prion diseases, Alzheimer’s disease, and Parkinson’s disease. In these diseases, protein misfolding and aggregation appear to expand through a process of seeded polymerization. Prion diseases occur in both humans and animals and are experimentally transmissible orally or by injection, thus providing a controllable model of other neurodegenerative protein misfolding diseases. In rodents and ruminants, prion disease has a slow course, lasting months to years. Although prion infectivity has been detected in brain tissue at 3 to 4 weeks postinfection (p.i.), the details of early prion replication in the brain are not well understood. Here we studied the localization and quantitation of PrPSc generation in vivo starting at 30 min postmicroinjection of scrapie into the brain. In C57BL mice at 3 days p.i., generation of new PrPSc was detected by immunohistochemistry and immunoblot assays, and at 7 days p.i., new generation was confirmed by real-time quaking-induced conversion assay. The main site of new PrPSc generation was near the outer basement membrane of small and medium blood vessels. The finding and localization of replication at this site so early after injection have not been reported previously. This predominantly perivascular location suggested that structural components of the blood vessel basement membrane or perivascular astrocytes might act as cofactors in the initial generation of PrPSc. The location of PrPSc replication at the basement membrane also implies a role for the brain interstitial fluid drainage in the early infection process.
IMPORTANCE Neurodegenerative diseases, including Alzheimer’s disease, Parkinson’s disease, and prion diseases, of humans are characterized by misfolding and aggregation of certain proteins, resulting in the destruction of brain tissue. In these diseases, the damage process spreads progressively within the central nervous system, but only prion diseases are known to be transmissible between individuals. Here we used microinjection of infectious prion protein (PrPSc) into the mouse brain to model early events of iatrogenic prion transmission via surgical instruments or tissue grafts. At 3 and 7 days postinjection, we detected the generation of new PrPSc, mostly on the outer walls of blood vessels near the injection site. This location and very early replication were surprising and unique. Perivascular prion replication suggested the transport of injected PrPSc via brain interstitial fluid to the basement membranes of blood vessels, where interactions with possible cofactors made by astrocytes or endothelia might facilitate the earliest cycles of prion infection.
“Early Generation of New PrPSc on Blood Vessels after Brain Microinjection of Scrapie in Mice” by Bruce Chesebro, James Striebel, Alejandra Rangel, Katie Phillips, Andrew Hughson, Byron Caughey, and Brent Race in mBio. Published online September 22 2015 doi:10.1128/mBio.01419-15