Summary: Creating modular neural networks (MoMNets), researchers generate a new way to study schizophrenia and other disorders in a lab setting.
Source: Columbia University
It was one of those coveted moments of serendipity in a scientific career.
Angeles Rabadan, Ph.D., left Raju Tomer’s lab at Columbia University for the day, but not before storing in an incubator a tiny tube with the few remaining drops of a cell-rich solution. When she returned the next morning, she saw something that made her eyes open wide.
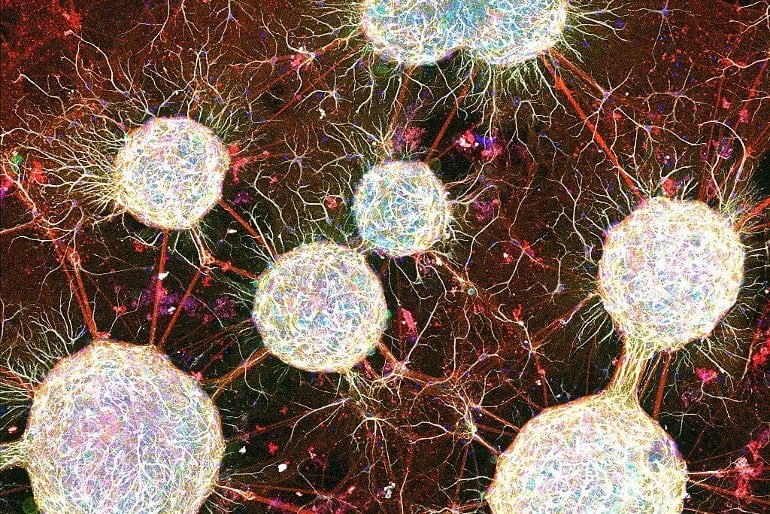
“Tiny balls had formed in the tube, and you could see there was something connecting them,” said Dr. Rabadan, an associate research neuroscientist in Columbia’s Department of Biological Sciences. Residual brain cells in the medium had aggregated overnight into minuscule neural spheres.
What’s more, these spheroids had spontaneously interconnected into a network. When she showed Dr. Tomer data from initial tests of the networks, the brain-like signaling he could see between the networks parts made him realize these networks could become a new and valuable research framework for neuroscience. The scientists dubbed these lab-grown renditions of brain-like cell assemblies Modular Neural Networks, or MoNNets.
What followed from Dr. Rabadan’s moment of serendipitous observation in the summer of 2017 has been a tantalizing research journey, now involving collaborators from at least eight Columbia departments and institutes.
The team’s research results so far, recently chronicled in the journal Nature Communications, stand a chance of helping the field of psychiatry medicine out of its long slump, the researchers and others say.
“For decades, there’s been very little progress in the treatment of psychiatric disorders,” said Joseph Gogos, MD, Ph.D., a principal investigator at Columbia’s Zuckerman Institute and one of the article’s authors.
“MoNNets could be a great help here because they’re well suited for both teasing out the biological bases of neurological and neuropsychiatric disorders and for efficiently vetting hundreds or thousands of possible pharmacological treatments,” added Gogos, who studies schizophrenia.
MoNNets are variations on an established laboratory theme in which researchers grow organoids: three-dimensional renditions of tissues and organs, some as big as a pea, housed in laboratory glassware. Scientists do this using precursor cells, stem cells, which they can nurture to develop into the various cell types of each organ.
Organoids have proven to be a powerful tool for studying organs such as the heart and liver. When it comes to the brain, however, organoids have not quite lived up to the initial hype, Dr. Tomer said, because they so far have lacked the network activity that allows different regions in actual brain tissue to communicate.
“MoNNets capture more of this network activity, which is why we think they will overcome the limitations of organoids and become better 3D models of brain tissue,” said Dr. Tomer, an assistant professor at Columbia and an Affiliate member of the Zuckerman Institute.
“With MoNNets, you have individual spheroid modules, which each do their own thing and perform their own computations, but each module also communicates through the entire system. That’s a very brain-like local-global dynamic. “
MoNNets are useful not just for simulating healthy brains but also for emulating diseased ones. In the new work, researchers grew MoNNets from mouse brain cells carrying gene variants associated with schizophrenia. The resulting MoNNets exhibited normal local communication between cells within spheroid modules, but poor coordination of signaling in the overall network of modules.
This lab-dish-to-disease correspondence suggests that MoNNets, comprising a few million cells, can mimic at least some of the cellular and neuron-circuitry features of brains of patients with schizophrenia comprising tens of billions of cells, Dr. Tomer said.
These same MoNNets embody another biological signature of brain circuits in schizophrenia. Their network structures and signaling behaviors are less stable compared to MoNNets grown with brain cells lacking the schizophrenia genes.
“The circuits and connections were changing and fluctuating,” said Dr. Tomer. “This could help us study short-term memory loss in schizophrenia, in which a similar absence of stable connections could be at play.”
MoNNets can list another resume item to strengthen their candidacy as petri-dish models of schizophrenia: When the researchers bathed the MoNNets with drugs known to bolster cognitive health in mice genetically engineered to emulate schizophrenia, the MoNNets’ cells and circuitry became more stable and exhibited more normal-looking network activity.
The team has already taken steps toward tailoring MoNNets for studying other neurological disorders, including epilepsy and Parkinson’s disease. Dr. Tomer envisions eventually developing a portfolio of tailored MoNNets that collectively model brain structures and functions associated with many neurological disorders.
“We could acquire data from these different disease models and then use data-analysis tools and machine learning to figure out what is biologically unique to each of these diseases,” says Dr. Tomer.
“In a systematic way we could model hundreds of genetic mutations, which are associated with these diseases, and use MoNNets to tease out how the different genes specifically affect brain circuits and health.” Data like this, Dr. Tomer and his colleagues say, would likely open the way to more accurate diagnoses and better treatment choices.
It also has the potential to shift the way scientists think about mental illness.
“We have long known from observations in animal and human brains that deficits at the neuronal-circuit level are prevalent in a range of psychiatric disorders, even though the genetic bases of these disorders are so varied,” said Dr. Gogos, who is also a professor of physiology, neuroscience and psychiatry at Columbia’s Vagelos College of Physicians and Surgeons.
Rather than characterizing mental illnesses as chemical imbalances in the brain that just need the right drug to be managed, the conceptual framework is shifting toward mental illness as a circuit disorder, Dr. Gogos said.
“What’s exciting about MoNNets is that they are opening new and versatile opportunities to model and study these circuit deficits in vitro in a dish,” he said.
Jordan Hamm, Ph.D., a neuroscientist at Georgia State University who was not among the authors of the Nature Communication paper, is an early fan of MoNNets. His own work in schizophrenia focuses on how mammalian brains process sensory information via interactions between local neural circuits and long-range, global brain networks.
“MoNNets represent a significant advance in tools for biomedicine and drug discovery, particularly for treatments aimed at pathologies of neural circuits and networks,” said Dr. Hamm. “These pathologies include some of the most devastating and still under-treated diseases, among them schizophrenia, autism and depression.”
About this schizophrenia research news
Author: Press Office
Source: Columbia University
Contact: Press Office – Columbia University
Image: The image is credited to Tomer lab / Columbia’s Dept. of Biological Sciences
Original Research: Open access.
“An in vitro model of neuronal ensembles” by M. Angeles Rabadan et al. Nature Communications
Abstract
An in vitro model of neuronal ensembles
Advances in 3D neuronal cultures, such as brain spheroids and organoids, are allowing unprecedented in vitro access to some of the molecular, cellular and developmental mechanisms underlying brain diseases.
However, their efficacy in recapitulating brain network properties that encode brain function remains limited, thereby precluding development of effective in vitro models of complex brain disorders like schizophrenia.
Here, we develop and characterize a Modular Neuronal Network (MoNNet) approach that recapitulates specific features of neuronal ensemble dynamics, segregated local-global network activities and a hierarchical modular organization. We utilized MoNNets for quantitative in vitro modelling of schizophrenia-related network dysfunctions caused by highly penetrant mutations in SETD1A and 22q11.2 risk loci.
Furthermore, we demonstrate its utility for drug discovery by performing pharmacological rescue of alterations in neuronal ensembles stability and global network synchrony. MoNNets allow in vitro modelling of brain diseases for investigating the underlying neuronal network mechanisms and systematic drug discovery.







