Summary: A new mouse study reveals a small cluster of interneurons in the hippocampus help the brain remember the places that matter the most. The findings could have implications for future studies of psychiatric disorders.
Source: Zuckerman Institute.
From the cab driver heading for Times Square to the commuter returning home on the freeway, we all carry maps in our head labeled with important locations. And a new Columbia study in mice shows that, by directing the delicate ebb-and-flow of brain activity, a small cluster of cells helps the brain’s internal GPS remember which places matter most. These findings underscore the fact that navigating an environment requires flexibility in the brain: brain-cell activity must change as memories are formed or recalled.
This research, published today in Neuron, could also provide insight into psychiatric disorders, such as autism and schizophrenia, which are often characterized by disruptions to this flexibility.
“Memories are fluid, not fixed, but observing precisely how brain cells, or neurons, flexibly lay down or recall memories has long proven challenging to scientists,” said Attila Losonczy, MD, PhD, a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper’s senior author. “With today’s study we provide, for the first time, visual evidence that a particular type of neurons makes this flexibility possible.”
The brain’s headquarters for learning and memory is the hippocampus, and it can be divided into distinct areas that process memory-related information. For this study, the researchers focused on area CA1, which encodes an animal’s location — as discovered by researchers who won the 2014 Nobel Prize.
In 2016, Dr. Losonczy’s lab found that CA1 neurons can act like a homing beacon; when a mouse looked for something, like water, neural activity spiked as the animal got close.
“The question then became: Was something directing this spike in activity?” said Dr. Losonczy, who is also a professor of neuroscience at Columbia’s Vagelos College of Physicians and Surgeons.
Broadly speaking, scientists divide neurons into two types: excitatory and inhibitory. Excitatory neurons are the gas pedal; they drive the activity of other neurons. Inhibitory neurons, by contrast, are the brakes: they suppress neural activity.
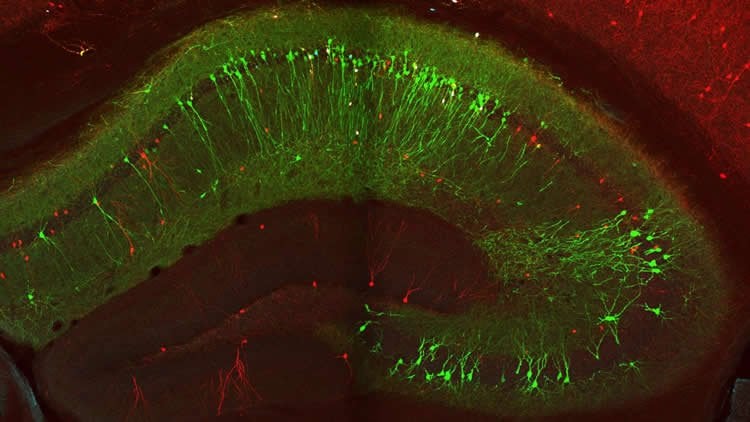
Today’s study focused on a particular type of CA1 inhibitory neurons called vasoactive intestinal polypeptide-expressing, or VIP, cells. Researchers had previously confirmed the existence of VIP cells, but had not examined them in the hippocampus of animals as they learned.
Using a two-photon microscope, Dr. Losonczy and his team monitored the VIP-cell activity as the mice ran on treadmills laden with various sights and sounds, some familiar and others new. This allowed the researchers to examine how the animals’ brains responded as they explored their surroundings, without a particular goal in mind.
In second set of experiments, the mice were given a task: find a water reward that had been placed at a specific, unmarked location along the treadmill’s path.
VIP-cell activity tended to spike during both sets of experiments: first as the animal ran aimlessly and then during goal-oriented learning when it sought the reward. Additional experiments and computational modeling, undertaken in collaboration with co-senior author Panayiota Poirazi, PhD, revealed how VIP cells heavily influenced CA1 neural activity.
“By default, CA1 excitatory neurons are kept ‘off,’ by a cluster of neighboring inhibitory neurons that target and suppress their activity,” said Dr. Losonczy. “But as the animals learned, a second cluster of inhibitory neurons — the so-called VIP cells — sprung to life.”
In a sequence of Rube Goldberg machine-like steps, the VIP cells first targeted the cluster of inhibitory neurons, effectively silencing them.
“With the inhibitory neurons no longer suppressing their activity, the excitatory neurons were then free to switch on,” said Dr. Losonczy. “This set off a chain reaction; activating the entire CA1 memory circuit and, ultimately, allowing the animals to learn.
As a reflection of this stepwise mechanism, Dr. Losonczy classifies VIP cells as disinhibitory neurons, because they act by inhibiting the inhibitory neurons. Disinhibition thus appears to be an ingenious way in which excitatory neurons can — in a roundabout way — be activated.
Though disinhibition may seem counterintuitive, Dr. Losonczy argues that it is actually illustrative of the delicate and fine-tuned nature of learning. A simple circuit — with one on-off switch — would be too blunt. An added layer of complexity may have evolved to match the complex nature of memory. This complexity also gives the circuit added flexibility when choosing which groups of neurons get activated, thus providing an additional, subtle way of adjusting itself during learning.
Moving forward, the research team will continue investigating VIP cells, as well as other cell types in the hippocampus involved in learning. They are also optimistic about what these findings mean for understanding disorders in which disrupted neural circuitry has been implicated.
“Normal brain function requires a delicate balance of excitation and inhibition, but many neuropsychiatric disorders, such as schizophrenia and autism, are characterized by an imbalance in this type of neural activity,” said Dr. Losonczy. “Our work, and the work of others, could very well help to elucidate how these disruptions result in these disorders’ devastating symptoms.”
Funding: This research was supported by the National Institute of Mental Health (1R01MH100631, 1U19NS104590, 1R01NS094668, F32MH18716, U01NS094330, U01NS099720), the Zegar Family Foundation, New York State Stem Cell Science (NYSTEM-C029157, S10OD0184464), the Revson Foundation and the ERC Starting Grant dEMORY (ERC-2012-StG-311435).
The authors report no financial or other conflicts of interest.
Source: Anne Holden – Zuckerman Institute
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Losonczy lab/Columbia’s Zuckerman Institute.
Original Research: Abstract for “Vasoactive Intestinal Polypeptide-Expressing Interneurons in the Hippocampus Support Goal-Oriented Spatial Learning” by Attila Losonczy et al. in Neuron. Published January 31 2019.
doi:10.1016/j.neuron.2019.01.009
[cbtabs][cbtab title=”MLA”]Zuckerman Institute”Pinpointing Cells That Control the Brain’s Memory Flow.” NeuroscienceNews. NeuroscienceNews, 31 January 2019.
<https://neurosciencenews.com/memory-flow-neurons-10671/>.[/cbtab][cbtab title=”APA”]Zuckerman Institute(2019, January 31). Pinpointing Cells That Control the Brain’s Memory Flow. NeuroscienceNews. Retrieved January 31, 2019 from https://neurosciencenews.com/memory-flow-neurons-10671/[/cbtab][cbtab title=”Chicago”]Zuckerman Institute”Pinpointing Cells That Control the Brain’s Memory Flow.” https://neurosciencenews.com/memory-flow-neurons-10671/ (accessed January 31, 2019).[/cbtab][/cbtabs]
Abstract
Vasoactive Intestinal Polypeptide-Expressing Interneurons in the Hippocampus Support Goal-Oriented Spatial Learning
Diverse computations in the neocortex are aided by specialized GABAergic interneurons (INs), which selectively target other INs. However, much less is known about how these canonical disinhibitory circuit motifs contribute to network operations supporting spatial navigation and learning in the hippocampus. Using chronic two-photon calcium imaging in mice performing random foraging or goal-oriented learning tasks, we found that vasoactive intestinal polypeptide-expressing (VIP +), disinhibitory INs in hippocampal area CA1 form functional subpopulations defined by their modulation by behavioral states and task demands. Optogenetic manipulations of VIP + INs and computational modeling further showed that VIP + disinhibition is necessary for goal-directed learning and related reorganization of hippocampal pyramidal cell population dynamics. Our results demonstrate that disinhibitory circuits in the hippocampus play an active role in supporting spatial learning.






