Summary: Synaptic pruning occurs as a result of mechanical tearing, a new study reveals.
Source: University of Munster
Nerve cells communicate with one another via long processes known as axons and dendrites, or, more generally, neurites. During development, these processes first grow and form connections with other cells, for example synapses with other nerve cells. Any neurites which are not properly linked, or are no longer needed, are removed by a corrective mechanism known as “pruning”.
Such pruning processes can appear drastic, and neurites sometimes seem to be severed directly from the nerve cell. Researchers headed by Dr. Sebastian Rumpf from the Institute of Neuro- and Behavioural Biology at Münster University (Germany) have now found the mechanism of neurite severing.
In a study published in the Journal of Cell Biology, the team show that in sensory nerve cells of the fruit fly Drosophila melanogaster, pruning occurs through mechanical tearing.
In the fruit fly, a large number of neurites are removed through pruning during the pupal phase, or metamorphosis.
“First the neurites become very unstable. This is due to an intrinsic mechanism.” says Sebastian Rumpf.
The main phase of pruning coincides with the phase where the animal changes its body shape, which involves strong body contractions.
“We have now discovered that these movements place such a stress on the fragile neurites that they are torn off,” Rumpf explains.
The severed neurites are then recognised as “waste” and removed by the surrounding tissue.
Animal cells constantly experience mechanical forces, to which they can react, for example, by changing their shape.
“Surprisingly little is known about the role of mechanical forces in the nervous system,” say lead authors Dr. Rafael Krämer and Dr. Neele Wolterhoff.
“Our work now shows how mechanical pulling can impact nerve cells.”
The pruning mechanism was previously unclear, and the role played by mechanical forces had not been investigated. Instead, experts suspected that there had to be a severing mechanism in the neurites themselves.
However, effects caused by potential “severing” enzymes could always be better explained in different ways, such that the mechanism remained unclear.
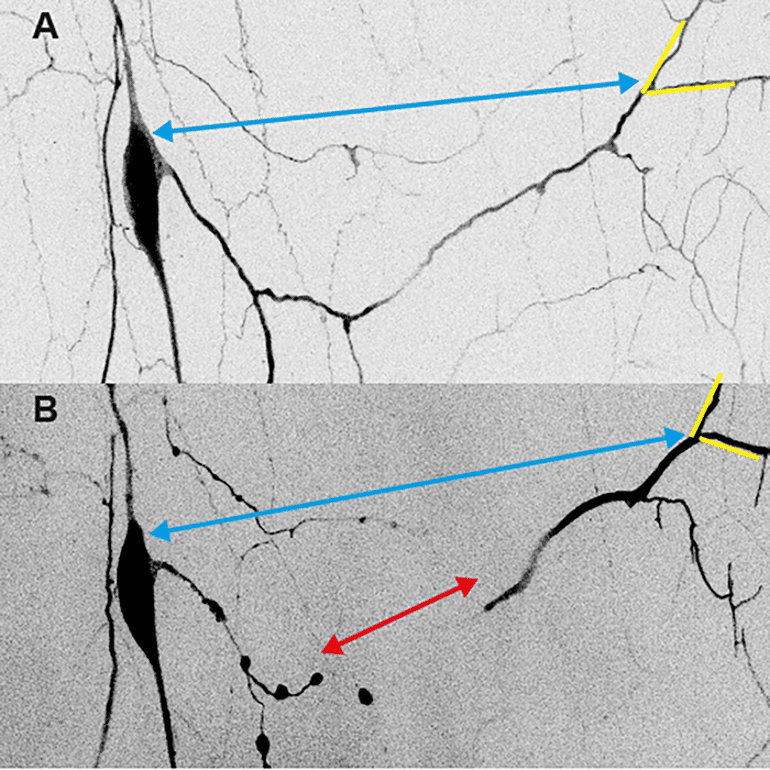
Demonstrating mechanical forces in Drosophila pupae is difficult because the animals cannot be removed from their pupal cases to take force measurements. In order to overcome this problem, Krämer and Wolterhoff followed the pruning process using live video microscopy, taking many long videos of pruning nerve cells. They then demonstrated the existences of pulling forces by analysis of these video recordings.
For example, the neurites initially became stretched before they were torn off, indicating that they were under tension. Using genetic manipulations, they could also decrease the morphogenetic movements, which caused defects in the pruning process.
About this neuroscience research news
Author: Christina Hoppenbrock
Source: University of Munster
Contact: Christina Hoppenbrock – University of Munster
Image: The image is credited to University of Münster – Rumpf Lab
Original Research: Open access.
“Developmental pruning of sensory neurites by mechanical tearing in Drosophila” by Sebastian Rumpf et al. Journal of Cell Biology
Abstract
Developmental pruning of sensory neurites by mechanical tearing in Drosophila
Mechanical forces actively shape cells during development, but little is known about their roles during neuronal morphogenesis. Developmental neurite pruning, a critical circuit specification mechanism, often involves neurite abscission at predetermined sites by unknown mechanisms.
Pruning of Drosophila sensory neuron dendrites during metamorphosis is triggered by the hormone ecdysone, which induces local disassembly of the dendritic cytoskeleton. Subsequently, dendrites are severed at positions close to the soma by an unknown mechanism.
We found that ecdysone signaling causes the dendrites to become mechanically fragile. Severing occurs during periods of increased pupal morphogenetic tissue movements, which exert mechanical forces on the destabilized dendrites.
Tissue movements and dendrite severing peak during pupal ecdysis, a period of strong abdominal contractions, and abolishing ecdysis causes non-cell autonomous dendrite pruning defects.
Thus, our data establish mechanical tearing as a novel mechanism during neurite pruning.






