Scientists find the brain works to minimize loss of vision, other functions.
A new study may have unlocked understanding of a mysterious part of the brain — with implications for neurodegenerative conditions such as Alzheimer’s. The results, published in Translational Vision Science & Technology (TVST), open up new areas of research in the pursuit of neuroprotective therapies.
Glaucoma is a neurodegenerative disease where patients lose seemingly random patches of vision in each eye. This random pattern of vision loss is in stark contrast to loss from a brain tumor or stroke, which causes both eyes to develop blind spots in the same location. Scientists have long thought that glaucoma’s progression is independent of – or uncontrolled by – the brain.
Last year, researchers found evidence that the progression of glaucoma is not random and that the brain may be involved after all. Specifically, they found patients with moderate to severe glaucoma maintained vision in one eye where it was lost in the other — like two puzzle pieces fitting together (a “Jigsaw Effect”). “This suggests some communication between the eyes must be going on and that can only happen in the brain,” explains the study’s lead author, William Eric Sponsel, MD, of the University of Texas at San Antonio, Department of Biomedical Engineering.
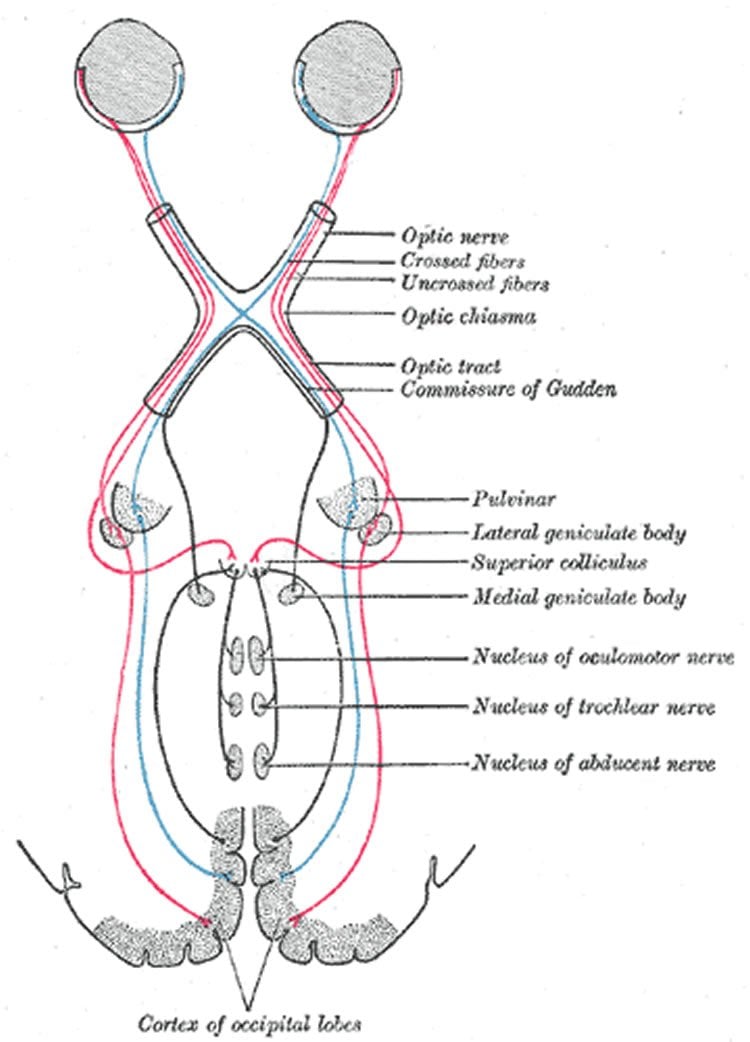
In the latest TVST paper, Refined Frequency Doubling Perimetry Analysis Reaffirms Central Nervous System Control of Chronic Glaucomatous Neurodegeneration, Sponsel and his research team found that the Jigsaw Effect begins at the earliest stages of glaucoma and discovered clues as to which part of the brain is responsible for optimizing vision in the face of glaucoma’s slow destruction of sight.
However, these findings, which challenge longstanding assumptions about glaucoma, have been met with skepticism. Other glaucoma experts challenged the results in a letter to the TVST editor. “If the brain controls the distribution of vision loss in glaucoma, then a patient’s vision with their two eyes should be better than if you simply ‘mix and match’ the vision of right and left eyes from different patients,” explained letter co-author Paul Artes, PhD, of Plymouth University, Department of Eye and Visual Sciences. Along with co-author Jonathan Denniss, PhD, University of Nottingham, Visual Neuroscience Group, their letter analyzed a new cohort of glaucoma patients in which “that’s essentially what we did. And we did not find any visual advantage in a patient’s own eyes versus the combined vision in eyes from different patients; indeed we found the opposite effect.”
Sponsel and co-authors responded to the letter to the editor with their own. “Our analysis of the data [Artes and Denniss] introduced demonstrated conclusively that the ‘Jigsaw Effect’ was indisputably present in patients we had never even seen. Moreover, we were able to confirm that the alternative analytical method they proposed could not reliably detect very obvious computer-generated complementary visual field pairs,” like a left and right eye that could only see opposite halves of their normal field of vision, says Sponsel. “The problem with their approach was their assumption that a single brain could somehow combine information from the eyes of different human beings. We studied individual people with naturally paired eyeballs connected to a single brain.”
The key to finding where the brain coordinates vision loss was found in small-scale, arc-shaped patterns of vision displayed by patients. Co-author Ted Maddess, PhD, of the Australian National University, Center of Excellence in Vision Science, explains that these patterns mimic structures found at the very back of the brain, known as ocular dominance columns. While their function is not completely understood, what is known is that some ocular dominance columns are associated with the left eye and other columns with the right.
The new paper suggests that the narrow spaces between ocular dominance columns associated with the left and right eye are where the brain coordinates each eye’s working field of vision. Depending on what the brain needs, those narrow spaces can function with either eye “much like a bilingual person living near the border of two countries,” explains Sponsel.
The progression of Alzheimer’s and Parkinson’s diseases, which have neurodegenerative biology similar to glaucoma, may also be actively mediated by the brain. “Our work has illustrated that the brain will not let us lose control of the same function on both sides of the brain if that can be avoided. It seems likely that the same kind of protective mechanism will be at work with other neurodegenerative disorders,” he says.
The investigative team believes that if the brain regulates neurodegeneration – that if the brain controls how it loses control – then researchers will now be able to look into largely unexplored regulatory processes for opportunities to slow or stop the progression of these diseases.
“We’ve opened up this beautiful new world; there is so much to discover here,” says Sponsel.
Additional Information: The letter to the editor “Extraordinary Claims Require Extraordinary Evidence: Centrally Mediated Preservation of Binocular Visual Field in Glaucoma is Unlikely” by Jonathan Denniss and Paul H. Artes is available here.
The response to the above letter “Definitive Response to Denniss and Artes: The Paired Eyes and Brain in One Person Are One Unit” by William E. Sponsel; Matthew A. Reilly; and Ted Maddess is available here.
Source: Katrina Norfleet – ARVO
Image Credit: The image is in the public domain
Original Research: Full open access research for “Refined Frequency Doubling Perimetry Analysis Reaffirms Central Nervous System Control of Chronic Glaucomatous Neurodegeneration” by Matthew A. Reilly; Analaura Villarreal; Ted Maddess; and William Eric Sponsel in Translational Vision Science & Technology. Published online June 2015 doi:10.1167/tvst.4.3.7
Abstract
Refined Frequency Doubling Perimetry Analysis Reaffirms Central Nervous System Control of Chronic Glaucomatous Neurodegeneration
Purpose:: Refined analysis of frequency doubling perimetric data was performed to assess binocular visual field conservation in patients with comparable degrees of bilateral glaucomatous damage, to determine whether unilateral visual field loss is random, anatomically symmetric, or non-random in relation to the fellow eye.
Methods:: Case control study of 41 consecutive patients with bilaterally mild to severe glaucoma; each right eye visual field locus was paired with randomly-selected co-isopteric left eye loci, performing 690,000 (10,000 complete sets of 69 loci) such iterations per subject. The potential role of anatomic symmetry in bilateral visual field conservation was also assessed by pairing mirror-image loci of the right- and left-eye fields. The mean values of the random co-isopteric and the symmetric mirror pairings were compared with natural point-for-point pairings of the two eyes by paired t-test.
Results:: Mean unilateral Matrix threshold across the entire 30-degree visual field were 17.0 dB left and 18.4 dB right (average 17.7). The better of the naturally paired concomitant loci yielded binocular equivalent mean bilateral Matrix threshold of 20.9 dB, 1.6 dB higher than the population mean of the 690,000 coisopteric pairings (t = −10.4; P < 10-12). Thus, a remarkable natural tendency for conservation of the binocular Matrix visual field was confirmed, far stronger than explicable by random chance. Symmetric pairings of precise mirror-image loci also produced values higher than random co-isopteric pairings (Δ 1.1 dB; t = −4.0; P = 0.0004).
Conclusions:: Refined data analysis of paired Matrix visual fields confirms the existence of a natural optimization of binocular visual function in severe bilateral glaucoma via interlocking fields that could only be created by CNS involvement. The disparity of paired Matrix threshold values at mirror-image loci was also highly nonrandom and quantitatively inverse from the expected if anatomic symmetry factors were merely passively contributing systematically to the compensatory binocular Matrix effect.
Translational Relevance:: The paired eyes and brain are reaffirmed to function as a unified system in the progressive age-related neurodegenerative condition chronic open angle glaucoma, maximizing the binocular visual field. Given the extensive homology of this disorder with other age-related neurodegenerations, it is reasonable to assume that the brain will similarly resist simultaneous bilateral loss of paired functional zones in both hemispheres in diseases like Alzheimer’s and Parkinson’s disease. Glaucomatous eyes at all stages of the disease appear to provide a highly accessible paired-organ study model for developing therapeutics to optimize conservation of function in neurodegenerative disorders.
“Refined Frequency Doubling Perimetry Analysis Reaffirms Central Nervous System Control of Chronic Glaucomatous Neurodegeneration” by Matthew A. Reilly; Analaura Villarreal; Ted Maddess; and William Eric Sponsel in Translational Vision Science & Technology. Published online June 2015 doi:10.1167/tvst.4.3.7