Summary: Studies in fish and mice found genome-wide changes in the gene expression of patterns in the skin, brains and liver following exposure to white fluorescent light. Both the skin and brains exhibited increased inflammation and immune response following exposure.
Source: Texas State University
Fluorescent lighting has become one of the most common artificial light sources in use today, but new research from Texas State University suggests there may be unexpected consequences at the genetic level.
The research team, comprised of Mikki Boswell, Yuan Lu, William Boswell, Markita Savage, Raquel Salinas and Ronald B. Walter from the Xiphophorus Genetic Stock Center in the Department of Chemistry and Biochemistry at Texas State, along with Kim Hildreth and Christi Walter from the Department of Cell Systems and Anatomy at the University of Texas Health San Antonio, have published their findings, “Fluorescent Light Incites a Conserved Immune and Inflammatory Genetic Response within Vertebrate Organs,” in the online journal Genes.
“Evolution occurred over many millennia exclusively under the full spectrum of sunlight. Thus, life had the opportunity to conscript each wavelength in the solar spectrum for the regulation of specific gene expression pathways,” Walter explained.
“Over the past 60 years, we have increasingly relied on artificial light sources that emit much narrower wavelength spectrums than does the sun. Yet, little research has been conducted to determine gene expression consequences, if any, from the use of common artificial light sources.”
In research using zebrafish (Danio rerio), Japanese rice fish (Oryzias latipes) and the hairless mouse (Mus musculus), the Texas State team identified genes activated by the wavelengths emitted by fluorescent light. The results show increased inflammation in tissue and organs in the subject animals, regardless of whether the species is primarily active in the day or night.
“In this report, we show genome-wide changes of gene expression patterns in skin, brain and liver for two commonly utilized fish experimental models (zebrafish and Japanese rice fish, also known as medaka), and a mammalian (mice), following exposure to 4,100 K ‘cool-white’ fluorescent light,” Walter said. “In spite of the extreme divergence of these animals (i.e., estimated divergence of mice and fish about 450 million years), and drastically different lifestyles (i.e., diurnal fish and nocturnal mice), the same highly conserved primary genetic response that involves activation of inflammation and immune pathways as part of an overall acute phase response was observed in the skin, brain and liver of all three animals. Follow-up studies to further define this response in mice are underway.”
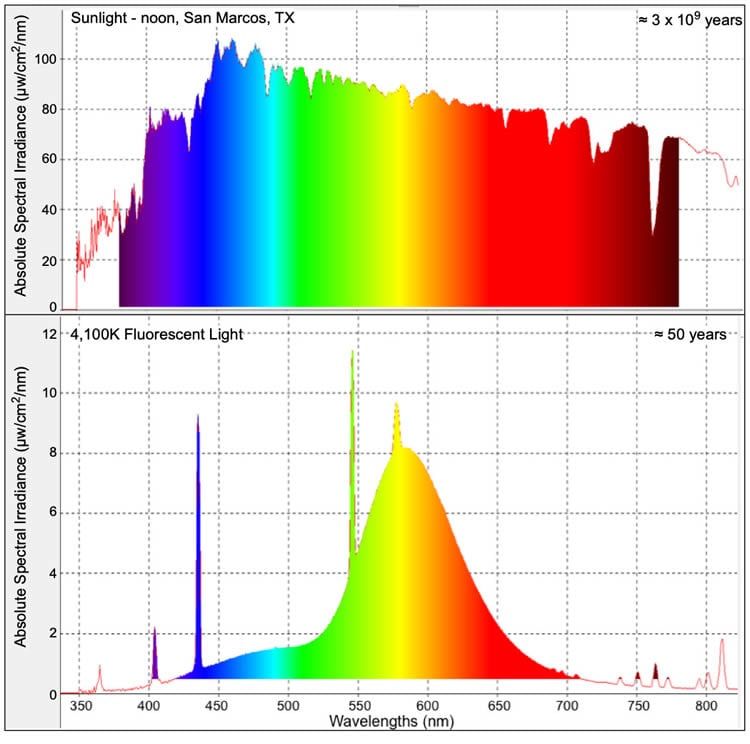
The team now hypothesizes that under broad-spectrum daylight conditions, these genes would be subsequently deactivated by wavelengths from a different part of the spectrum — but these additional wavelengths may be absent in fluorescent lighting.
The skin and brain of the three animals, as well as the liver of both fish models all exhibit, increased inflammation and immune responses; however, the mouse liver suppressed this reaction. Overall, the conserved nature of the genetic responses observed, among fishes and a mammal, suggest the presence of light responsive genetic circuitry that is deeply embedded in the vertebrate genome.
Source:
Texas State University
Media Contacts:
Jayme Blaschke – Texas State University
Image Source:
The image is credited to the researchers.
Original Research: Open access.
“Fluorescent Light Incites a Conserved Immune and Inflammatory Genetic Response within Vertebrate Organs (Danio rerio, Oryzias latipes and Mus musculus)”
Mikki Boswell, Yuan Lu, William Boswell, Markita Savage, Kim Hildreth, Raquel Salinas, Christi A. Walter and Ronald B. Walter. Genes doi:10.3390/genes10040271
Abstract
Fluorescent Light Incites a Conserved Immune and Inflammatory Genetic Response within Vertebrate Organs (Danio rerio, Oryzias latipes and Mus musculus)
Fluorescent light (FL) has been utilized for ≈60 years and has become a common artificial light source under which animals, including humans, spend increasing amounts of time. Although the solar spectrum is quite dissimilar in both wavelengths and intensities, the genetic consequences of FL exposure have not been investigated. Herein, we present comparative RNA-Seq results that establish expression patterns within skin, brain, and liver for Danio rerio, Oryzias latipes, and the hairless mouse (Mus musculus) after exposure to FL. These animals represent diurnal and nocturnal lifestyles, and ≈450 million years of evolutionary divergence. In all three organisms, FL induced transcriptional changes of the acute phase response signaling pathway and modulated inflammation and innate immune responses. Our pathway and gene clustering analyses suggest cellular perception of oxidative stress is promoting induction of primary up-stream regulators IL1B and TNF. The skin and brain of the three animals as well as the liver of both fish models all exhibit increased inflammation and immune responses; however, the mouse liver suppressed the same pathways. Overall, the conserved nature of the genetic responses observed after FL exposure, among fishes and a mammal, suggest the presence of light responsive genetic circuitry deeply embedded in the vertebrate genome.







